
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
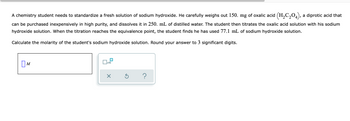
Transcribed Image Text:A chemistry student needs to standardize a fresh solution of sodium hydroxide. He carefully weighs out 150. mg of oxalic acid (H₂C₂O4), a diprotic acid that
can be purchased inexpensively in high purity, and dissolves it in 250. mL of distilled water. The student then titrates the oxalic acid solution with his sodium
hydroxide solution. When the titration reaches the equivalence point, the student finds he has used 77.1 mL of sodium hydroxide solution.
Calculate the molarity of the student's sodium hydroxide solution. Round your answer to 3 significant digits.
x10
M
X
5
?
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by stepSolved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- An analytical chemist weighs out 0.193 g of an unknown diprotic acid into a 250 mL volumetric flask and dilutes to the mark with distilled water. She then titrates this solution with 0.1200 M NaOH solution. When the titration reaches the equivalence point, the chemist finds she has added 24.0 mL of NaOH solution. Calculate the molar mass of the unknown acid. Be sure your answer has the correct number of significant digits. g mol x10 X Śarrow_forwardYou have been provided with a 2.08 x 10-2 L sample of lithium hydroxide (LiOH) of unknown concentration. You perform a titration with 2.19 M nitric acid (HNO3), and find that 24.6 mL are required to reach the equivalence point (as determined using a coloured indicator). What is the concentration of the lithium hydroxide solution in mol L-1?arrow_forwardA chemist needs to determine the concentration of a solution of nitric acid, HNO3. She puts 905 mL of the acid in a flask along with a few drops of indicator. She then slowly adds 0.200 mol L Ba(OH)2 to the flask until the solution turns pink, indicating the equivalence point of the titration. She notes that 225 mL of Ba(OH)2 was needed to reach the equivalence point. Solution map In this titration, the concentration of base is known and can be used to calculate the unknown acid concentration: concentration of base → moles of base → moles of acid → concentration of acid Part A How many moles of Ba(OH)2 are present in 225 mL of 0.200 mol L-1 Ba(OH)2? Express your answer numerically in moles. • View Available Hint(s) Vo AEO ? mol Ba(OH)2 Submitarrow_forward
- An analytical chemist weighs out 0.181 g of an unknown diprotic acid into a 250 mL volumetric flask and dilutes to the mark with distilled water. She then titrates this solution with 0.1700 M NaOH solution. When the titration reaches the equivalence point, the chemist finds she has added 15.9 mL of NaOH solution. Calculate the molar mass of the unknown acid. Be sure your answer has the correct number of significant digits. ol. g mol Ararrow_forwardA chemistry student weighs out 0.159 g of ascorbic acid (H,C,H,0), a diprotic acid, into a 250. mL volumetric flask and dilutes to the mark with distilled water. He plans to titrate the acid with 0.0800M NaOH solution. Calculate the volume of NaOH solution the student will need to add to reach the final equivalence point. Be sure your answer has the correct number of significant digits. mLarrow_forwardYou have a 39.2 mL sample of hydrochloric acid (HCl) of unknown concentration. A titration is performed with 1.48 M sodium hydroxide (NaOH), and find 3.81 x 10-2 L are required to reach the equivalence point . What is the hydrochloric acid concentration?arrow_forward
- An analytical chemist weighs out 0.026 g of an unknown monoprotic acid into a 250 mL volumetric flask and dilutes to the mark with distilled water. She then titrates this solution with 0.0700 M NaOH solution. When the titration reaches the equivalence point, the chemist finds she has added 3.8 mL of NaOH solution. Calculate the molar mass of the unknown acid. Be sure your answer has the correct number of significant digits. g x10 molarrow_forwardAn analytical chemist weighs out 0.318 g of an unknown triprotic acid into a 250 mL volumetric flask and dilutes to the mark with distilled water. He then titrates this solution with 0.0600 M NaOH solution. When the titration reaches the equivalence point, the chemist finds he has added 82.8 mL of NaOH solution. Calculate the molar mass of the unknown acid. Be sure your answer has the correct number of significant digits. mol x10 x 3arrow_forwardA chemistry student weighs out 0.0856 g of citric acid (H,CH,0,), a triprotic acid, into a 250. mL volumetric flask and dilutes to the mark with distilled water. He plans to titrate the acid with 0.0900 M NaOH solution. Calculate the volume of NaOH solution the student will need to add to reach the final equivalence point. Be sure your answer has the correct number of significant digits. mL x10arrow_forward
- An analytical chemist weighs out 0.050 g of an unknown triprotic acid into a 250 mL volumetric flask and dilutes to the mark with distilled water. He then titrates this solution with 0.1600 M NaOH solution. When the titration reaches the equivalence point, the chemist finds he has added 9.6 mL of NaOH solution. Calculate the molar mass of the unknown acid. Be sure your answer has the correct number of significant digits. g molarrow_forwardAn analytical chemist weighs out 0.185 g of an unknown monoprotic acid into a 250 mL volumetric flask and dilutes to the mark with distilled water. She then titrates this solution with 0.2000 M NAOH solution. When the titration reaches the equivalence point, the chemist finds she has added 9.5 mL of NaOH solution. Calculate the molar mass of the unknown acid. Be sure your answer has the correct number of significant digits. g x10 molarrow_forwardYou are preparing standard acid and base solutions for the laboratory, using potassium hydrogen phthalate (KHC₈H₄O₄, abbreviated KHP) as the primary standard. KHP (molar mass = 204.22 g/mol) has one acidic hydrogen. You prepared solutions of both NaOH and HCl. It took 22.65 mL of the NaOH solution to titrate (react exactly with) 1.55 g KHP. It then took 32.35 mL of HCl solution to titrate 25.00 mL of the NaOH solution. What is the molarity of the HCl solution?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY