
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
thumb_up100%
Please help 2 2
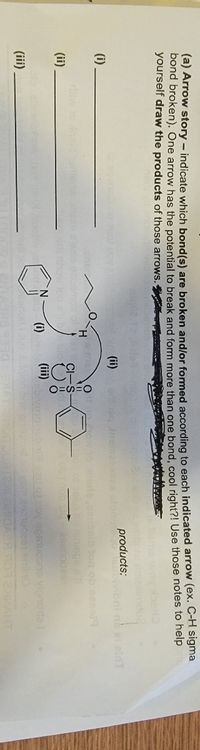
Transcribed Image Text:(a) Arrow story – indicate which bond(s) are broken and/or formed according to each indicated arrow (ex. C-H sigma
bond broken). One arrow has the potential to break and form more than one bond, cool right?! Use those notes to help
yourself draw the products of those arrows. P
products:
(ii)
ivibni n el einT
(i)
eher
Selb ton ob pasol9
H.
(ii)
2sanogae
(i)
(iii)
pcone vipnote
(iii)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution
Trending nowThis is a popular solution!
Step by stepSolved in 4 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Help pleasearrow_forwardA bucket of water has a volume of 0.473 kL . How many dL is this? STARTING AMOUNT ADD FACTOR ANSWER RESET *( ) 47300 1000 0.001 0.01 10 4730 0.0000473 100 0.473 0.1 1 0.00473 473 0.000473 kL dL +arrow_forward2:59 AT&T Aa 2:58 PM, Apr 22 Save Share II H3G H3C H30* ? Brz H3C SCH3arrow_forward
- STARTING AMOUNT esc C X C 2 F2 W # 3 1000 F3 ADD FACTOR x( ) $ 4 1 97.9 FS de in % 5 2 Convert 23.4 kJ to calories 2.34 x 104 t 4184 kcal 6 P 0.001 J 4.184 COMEC DOLL ANSWER & 7 9.79 × 104 cal 00 8 RESET FO 2 23.4 ( 5590 A FIT 120 Jan 21 del +arrow_forwardBa(NO,l(aq) + K,SOlaq) Baso(s) + 2 KNOlaq) Volume Volume Trial 0.10 M Ba(NO)2 (ml) 0.10 M K,SO, (ml) Mass Baso4 (s) Limiting Reagent 50. 20. 0.46 50. 40. 0.92 3 50. 50. 1.15 4 50. 60. 1.15 50. 80. 1.15 • Question 8 Use the data to identify the limiting reagent for each reaction performed. V Trail 1 a. Neither/Both EV Trial 2 b. Baso4 EV Trial 3 C. KSO4 EV Trial 4 d. Ba(NO3)2 EV Trial 5 e. KNO3 Question Help: Message instructor I Calculator Submit Question • Question 9 Why is the amount of precipitate in the Data Table above the same in Trials 3 through 5? Edit Insert Formats B IVX x' Aarrow_forwardConcentration (M) 1 0.8 0.6 0.4 0.2 0 0 1st attempt 10 y = -0.0111x + 1.0362 20 30 Time (minutes) What is the y-intercept of this graph? 40 50 60arrow_forward
- 4-16. Zinc is an essential micronutrient in pet food, but is toxic if present in excess. Do the Zn concentrations (mg/g) for the five cat foods and two dog foods determined by two methods differ significantly at the 95% confidence level? Cat 1 Cat 2 Cat 3 Cat 4 Cat 5 Dog 1 Dog 2 Old method: bris 8 Я 84.9 73.5 173.0 62.7 154.0 80.1 185.0 New faster method: 86.2 81.8 186.0 73.4 138.0 72.5 203.0 SO Souza, S. S. L. Costa, R. G. O. Araujo, C. A. Cats andarrow_forwardNeed help with homeworkarrow_forward10 11 12 13 14 15 16 17 18 19 20 Calculate the solubility of BaCro, in water at 25 °C. You'll find K data in the ALEKS Data tab. sp Round your answer to 2 significant digits. Continue MacBook Airarrow_forward
- A Aa A Y F 3 States) Text Predictions: On 13 C 14 101 !!! M $ 4 ET 垣~ Paragraph What is the difference between a cylinder and a test tube? B 1.The cylinder is made of glass and the test tube is made of glass. f5 Accessibility: Good to go % 5 3.The cylinder is the same as the test tube, it's just another name for it. V 2.The cylinder contains dimensions to measure volume and the test tube is to observe reactions. f6 4.The test tube comes in a single measure (25 ml) and the test tube can come in different volumes (25- 200 ml for example). 5 A 6 P 17 Normal & 7 18 7 hp No Spacing O 19 Styles 8 Heading 1 f10 f17 .. V F12 5 فکر Ed 82°F Mosarrow_forwardCHM 431A Lab 7- 2021SP Given Data page Time Temperatu (min) C1. Boiling temp of water re (°C) 25.4 Initial temperature of water 32.5 Temperature of boiling water 44.9 3. 56.9 C2. Temperature change (AT) 4. 71.3 Mass of beaker 5. 86.6 100.2 Mass of beaker and water 7. 100.6 C3. Mass of water 8. 100.6 100.6 1. 2) 6.arrow_forwardOne of the most important applications of ion exchange is the separation of large molecules from small molecules? 1- true 2- Errorarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY