
Elements Of Electromagnetics
7th Edition
ISBN: 9780190698614
Author: Sadiku, Matthew N. O.
Publisher: Oxford University Press
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Note:-
• Do not provide handwritten solution. Maintain accuracy and quality in your answer. Take care of plagiarism.
• Answer completely.
• You will get up vote for sure.
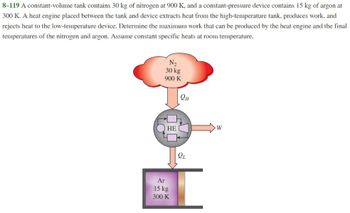
Transcribed Image Text:8-119 A constant-volume tank contains 30 kg of nitrogen at 900 K, and a constant-pressure device contains 15 kg of argon at
300 K. A heat engine placed between the tank and device extracts heat from the high-temperature tank, produces work, and
rejects heat to the low-temperature device. Determine the maximum work that can be produced by the heat engine and the final
temperatures of the nitrogen and argon. Assume constant specific heats at room temperature.
N₂
30 kg
900 K
HE
Ar
15 kg
300 K
QH
QL
W
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution
Trending nowThis is a popular solution!
Step by stepSolved in 4 steps with 4 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, mechanical-engineering and related others by exploring similar questions and additional content below.Similar questions
- Example: Thesis Statement (1) Check () the thesis statements that suggest a chronological order. Put a double check (V) next to the thesis statements that suggest the essay will describe a process or procedure. In the sentences you have checked, circle the word or words that indicate chronological order or process. к1. A child learns to handle responsibility in a series of small steps. X2. 3. WA. 5. X6. X7. X8. 9. A person's intelligence is the product of both heredity' and environment. There are two main reasons I believe women in the army should not be allowed in a war zone along with men. The procedure for submitting expense reports has recently changed. The tensions that led to last year's student riots had been developing for several years. North American directness often conflicts with Asian modesty. The two busiest travel days in the United States are the Wednesday before and the Sunday after Thanksgiving. Cultures celebrate the end of winter and the arrival of spring in…arrow_forwardB.How does the color of t-shirt you wear affect its temperature? te three variations of the independent variable below. IF THEN BECAUSE (Write a SPECIFIC action you can do) (Make a prediction about what you are measuring) (Reason why from your knowledge/research)arrow_forwardI need these three parts answered, if you are unable to answer all three parts please leave it for another tutor to answer, thank you.arrow_forward
- Don't need to show work, just need to know which ones correct to compare my answers. 7.8.arrow_forwardPlease make the charts for the questions. Please refer to Successful Project Management (7th Edition). Attached is the example Thank you.arrow_forward71.7 is being marked wrong in McGraw Hill homework. I thought I had done it right with putting 86.03 but that is wrong also. I'm not trying to just find the answer, trying to find how to do this. Could there be a different number than 71.7? Where does the μk=0.20 come into the equation?arrow_forward
- I have to chart this in excel and show my work if possible. Thank you in advance.arrow_forwardWhen interpolating which of the following will give you the best possible results? Select two points that are the closest to the unknown. Use interpolation only when the values follow a linear curve. Do not use interpolation for values that follow an exponential curve. Do not use interpolation for an unknown outside of known values. Do not use on values that are slowly varying.arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press
Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON
Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education
Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY
Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning
Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY
Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY

Elements Of Electromagnetics
Mechanical Engineering
ISBN:9780190698614
Author:Sadiku, Matthew N. O.
Publisher:Oxford University Press

Mechanics of Materials (10th Edition)
Mechanical Engineering
ISBN:9780134319650
Author:Russell C. Hibbeler
Publisher:PEARSON

Thermodynamics: An Engineering Approach
Mechanical Engineering
ISBN:9781259822674
Author:Yunus A. Cengel Dr., Michael A. Boles
Publisher:McGraw-Hill Education

Control Systems Engineering
Mechanical Engineering
ISBN:9781118170519
Author:Norman S. Nise
Publisher:WILEY

Mechanics of Materials (MindTap Course List)
Mechanical Engineering
ISBN:9781337093347
Author:Barry J. Goodno, James M. Gere
Publisher:Cengage Learning

Engineering Mechanics: Statics
Mechanical Engineering
ISBN:9781118807330
Author:James L. Meriam, L. G. Kraige, J. N. Bolton
Publisher:WILEY