
Introduction to Chemical Engineering Thermodynamics
8th Edition
ISBN: 9781259696527
Author: J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark Swihart
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Question
thumb_up100%
6.52
- Single pass and overall conversion: The diagram below shows a chemical process in which component A reacts to become B in the reactor, with reaction stoichiometry of A → 2B, followed by partial separation of A & B in a distillation tower, and recycle of stream 4 (high in unreacted A) back to stream 1. For the values given in the process stream table below, determine what is the single pass conversion and the overall conversion based upon component A.
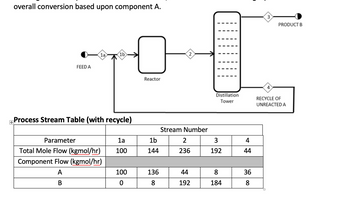
Transcribed Image Text:overall conversion based upon component A.
FEED A
1a
1b
Process Stream Table (with recycle)
Parameter
Total Mole Flow (kgmol/hr)
Component Flow (kgmol/hr)
A
B
1a
100
100
0
Reactor
1b
144
136
8
2
Stream Number
2
236
44
192
Distillation
Tower
3
192
8
184
4
44
36
8
3
PRODUCT B
RECYCLE OF
UNREACTED A
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution
Trending nowThis is a popular solution!
Step by stepSolved in 3 steps with 3 images

Follow-up Questions
Read through expert solutions to related follow-up questions below.
Follow-up Question
this was great but can you please re-do problem and rewrite equations in word, i have a hard time reading.
Solution
by Bartleby Expert
Follow-up Questions
Read through expert solutions to related follow-up questions below.
Follow-up Question
this was great but can you please re-do problem and rewrite equations in word, i have a hard time reading.
Solution
by Bartleby Expert
Knowledge Booster
Similar questions
- Propylene is produced by the dehydrogenation of propane. The product stream leaving a dehydrogenation reactor is found to contain 900 mol/s propane, 99.75 mol/s propylene, and 95 mol/s of H2. This stream is passed to a distillation column, with the distillate containing all of H2, most of propylene, and 0.555% of the molar flow rate of the propane that enters the column. The bottoms from the column, which is recycled back to the reactor, has a propylene molar flow rate equal to 5% of that in the distillate. The distillate is then passed to a second distillation column in which all of the H2 is recovered in the distillate and all of the organics (propane and propylene) are recovered in the bottoms. a) Construct a process flow diagram for this system.arrow_forwardThe formation of NO from Na and O is to be carried out in a small batch reactor. As a first approximation, we shall consider that the reaction time is more rapid than the time of cylinder compression, consequently, the reaction takes place isothermally in car eylinder at 2700 K, in a constant volume reactor (cylinder) and under a pressure of 20 atm, the initial concentration of N2 is 0.0696 mol/liter. By a specifying constant volume, we are assuming that reaction take place rapidly with respect to the movement of the piston in the cylinder. Consider that the feed consists of 77% N2, 15% Oz and 8% other gases, which may be considered inert. At this temperature the equilibrium constant (Ke-0.01). The reaction is reversible: Na + 0z + 2NO With a rate equation: Cho -TN, = k ( CN, Co, Ke Calculate: a) The equilibrium conversion of N2. b) The time required to achieve 80% of the equilibrium conversion. The formation reaction rate- constant k at this temperature is 1.11 liter mol.h' Hint: dx…arrow_forwardEthane (C2H6) gas at mass flow rate of 1.5 kg/min is mixed with air (O2 + 3.76N2) at mass flow rate of 1 kg/sec in a steady-state, steady-flow isobaric combustion process. All reactants enter the process at 298 K and 100 kPa, and the fuel burns to complete combustion. a) Determine the Air-Fuel Ratio (AFR) b) Write a balanced reaction equation based on one mole of fuel c) Determine the excess air fraction (%) d) Assuming that the combustion products exit the system at 1000 K, determine the volumetric flow rate of the combustion exhaust (m3 /s) e) Assuming that the combustion products exit the system at 1000 K, determine the rate of heat transfer to/from the system (kW)Repeat steps d and e, but assuming that the combustion product exit the system at 2000 k. Using the values in part e for 1000 k and 2000 k, apply linear interpolation to estimate the adiabatic exit temperature (?̇ = 0) for the combustion products. Next, using conservation of energy analysis, solve for the adiabatic exit…arrow_forward
- For the electrolysis of CuCl2 (molten) to form Cu(s) and Cl2(g):E°Cu2+/Cu = 0.339 VE°Cl-/Cl2 = 1.360 V What minimum voltage must be used to carry out the reaction? If 1.5V is used, how much electrical energy (in kJ) will be used to produce 2g of Cl2(s)?arrow_forwardgas phase reactionA -> B + Ccarried out isothermally in a constant volume batch reactor of 100 L. An amount of 100 moles of pure A is initially placed in the reactor. The reactor is well mixed and the ideal gas equation holds. If the reaction is first order,-rA = kCA with k = 1 1/sCalculate the time required to consume 80% (moles) of A.arrow_forwardQ4) The reaction A + B = C + D proceeds to equilibrium at a temperature of 573 K. The mole fractions of the four reactive species satisfy the equilibrium relation. The feed to a reactor contains 2.0 mol of A, 3.0 mol of B, 0.5 mol of C and no mol of D. If K (573) = 5, Calculate: a) The mole fractions of the final product. b) The fractional conversion of the limiting reactant.arrow_forward
- complete part E onlyarrow_forwardPlease help....arrow_forwardIn a batch reactor, a substance A was processed, which generated different products (D and U), through competitive parallel reactions with the following reaction kinetics: After 20 minutes of reaction, it was determined that the composition of the reaction medium was CA = 1 mol/L, CD = 5 mol/L, CU = 2 mol/L. The option that indicates, respectively, the instantaneous and global selectivities at the end of the reaction are: A) 3 and 2. B) 2.5 and 2. C) 2 and 3. D) 2 and 2.5.arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 Introduction to Chemical Engineering Thermodynami...Chemical EngineeringISBN:9781259696527Author:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark SwihartPublisher:McGraw-Hill Education
Introduction to Chemical Engineering Thermodynami...Chemical EngineeringISBN:9781259696527Author:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark SwihartPublisher:McGraw-Hill Education Elementary Principles of Chemical Processes, Bind...Chemical EngineeringISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...Chemical EngineeringISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY Elements of Chemical Reaction Engineering (5th Ed...Chemical EngineeringISBN:9780133887518Author:H. Scott FoglerPublisher:Prentice Hall
Elements of Chemical Reaction Engineering (5th Ed...Chemical EngineeringISBN:9780133887518Author:H. Scott FoglerPublisher:Prentice Hall
 Industrial Plastics: Theory and ApplicationsChemical EngineeringISBN:9781285061238Author:Lokensgard, ErikPublisher:Delmar Cengage Learning
Industrial Plastics: Theory and ApplicationsChemical EngineeringISBN:9781285061238Author:Lokensgard, ErikPublisher:Delmar Cengage Learning Unit Operations of Chemical EngineeringChemical EngineeringISBN:9780072848236Author:Warren McCabe, Julian C. Smith, Peter HarriottPublisher:McGraw-Hill Companies, The
Unit Operations of Chemical EngineeringChemical EngineeringISBN:9780072848236Author:Warren McCabe, Julian C. Smith, Peter HarriottPublisher:McGraw-Hill Companies, The

Introduction to Chemical Engineering Thermodynami...
Chemical Engineering
ISBN:9781259696527
Author:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark Swihart
Publisher:McGraw-Hill Education

Elementary Principles of Chemical Processes, Bind...
Chemical Engineering
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY

Elements of Chemical Reaction Engineering (5th Ed...
Chemical Engineering
ISBN:9780133887518
Author:H. Scott Fogler
Publisher:Prentice Hall


Industrial Plastics: Theory and Applications
Chemical Engineering
ISBN:9781285061238
Author:Lokensgard, Erik
Publisher:Delmar Cengage Learning

Unit Operations of Chemical Engineering
Chemical Engineering
ISBN:9780072848236
Author:Warren McCabe, Julian C. Smith, Peter Harriott
Publisher:McGraw-Hill Companies, The