
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
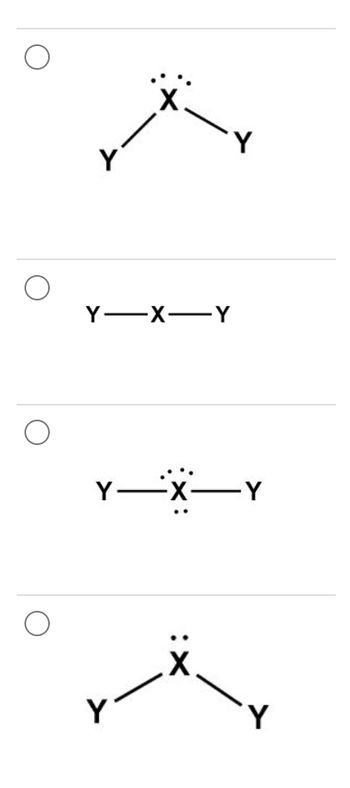
Transcribed Image Text:Y-X-Y
Y
-X-Y
X

Transcribed Image Text:6
What is the geometry for an
unknown molecule, XS2,
where X is an element with
4 valence electrons and an
electronegativity of 2.4
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution
Trending nowThis is a popular solution!
Step by stepSolved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 2. a. The Pauling scale of electronegativity is based upon bond dissociation energies, D, and involves using the equation DAB = (DAz × Dg2)"² + A . For what process does A represent the energy? How is A related to electronegativities? b. The electronegativities of H, S, and O are 2.2, 2.58, and 3.44 respectively. Explain the fact that the HOH bond angle in H,O is 104.5 ° while the HSH bond angle in H,S is only 92.1° i. based upon relative electronegativities and ii based upon atomic orbital hybridization.arrow_forwardThe Lewis symbol for the oxygen atom has valence electrons. The number of covalent bonds which oxygen usually forms in order to complete its valence shell and obey the octet rule is O 4, 4 O 6, 3 O 6, 2 O 6, 1arrow_forwardIn which of the following pairs is the first element expected to have a higher electronegativity than the second? O 0, P O Cs, Rb O I, Br Al, Parrow_forward
- Given the electronegativity for O is 3.5 and S is 2.5 in Sulfate, would you predict the molecule to be polar or nonpolar"arrow_forwardCarbon dioxide is a greenhouse gas. Which of the following is the correct Lewis structure for carbon dioxide? 0=c=0arrow_forwardDecide whether these proposed Lewis structures are reasonable. proposed Lewis structure -C=C:] H-C : 0: 0=0=0 [HO] : O OO Is the proposed Lewis structure reasonable? Yes. No, it has the wrong number of valence electrons. The correct number is: No, it has the right number of valence electrons but doesn't satisfy the octet rule. The symbols of the problem atoms are:* 0 Yes. No, it has the wrong number of valence electrons. The correct number is: No, it has the right number of valence electrons but doesn't satisfy the octet rule. The symbols of the problem atoms are:* П Yes. No, it has the wrong number of valence electrons. The correct number is: No, it has the right number of valence electrons but doesn't satisfy the octet rule. The symbols of the problem atoms are:* * If two or more atoms of the same element don't satisfy the octet rule, just enter the chemical symbol as many times as necessary. For example, if two oxygen atoms don't satisfy the octet rule, enter "O,0".arrow_forward
- Ll.59.arrow_forwardWhat is the difference between a simple covalent bond and a dative covalent bond?arrow_forwardWhen drawing the Lewis structure of the HCC13 molecule, the structure should represent a total of 26 valence electrons. What is the best description of the complete Lewis structure of the molecule? Select one: AH atom should be in the center with single bonds to each Cl atom and a double bond to the C atom. A Cl atom should be in the center with a single bond to the C atom and a single bond to the H atom. AC atom should be in the center with a double bond to the H atom and double bonds to each Cl atom. AC atom should be in the center with single bonds to each Cl atom and a single bond to the H atom.arrow_forward
- Decide whether these proposed Lewis structures are reasonable. proposed Lewis structure :Z: I :Z: c=0 0 0 0 Is the proposed Lewis structure reasonable? O Yes. O No, it has the wrong number of valence electrons. The correct number is: O O No, it has the right number of valence electrons but doesn't satisfy the octet rule. The symbols of the problem atoms are:* ■ Yes. No, it has the wrong number of valence electrons. The correct number is: No, it has the right number of valence electrons but doesn't satisfy the octet rule. The symbols of the problem atoms are:* O Yes. O No, it has the wrong number of valence electrons. The correct number is: No, it has the right number of valence electrons but doesn't satisfy the octet rule. The symbols of the problem atoms are:* * If two or more atoms of the same element don't satisfy the octet rule, just enter the chemical symbol as many times as necessary. For example, if two oxygen atoms don't satisfy the octet rule, enter "0,0". X 3arrow_forwardWhich one of the following bonds is the most polar?(electronegativities: C = 2.5, O = 3.5, Cl = 3.0, N = 3.0, H = 2.1)arrow_forward3. Carbon atoms will bond with each other to form a chain, often with the peripheral atoms being hydrogen. These compounds, hydrocarbons, are organic compounds composed only of carbon and hydrogen. However, when hydrogen atoms are substituted with other elements, such as fluorine, this changes the properties of the molecule. Methylfluoride (CH3F), a substituted hydrocarbon, is used in semiconductor processing. Figure 1 depicts an incorrect Lewis structure for CH3F. (QUestion shown in photo) Which reason best explains why this structure is incorrect? a) Hydrogen lacks an octet. b) Too few electrons are used. c) Carbon cannot exceed an octet. d) Fluorine atom has too many electrons.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY