
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
thumb_up100%
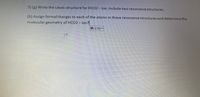
Transcribed Image Text:5) (a) Write the Lewis structure for HCO2 - ion. Include two resonance structures.
(b) Assign formal charges to each of the atoms in these resonance structures and determine the
molecular geometry of HCO2- ion?
B(Ctrl)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution
Trending nowThis is a popular solution!
Step by stepSolved in 3 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Consider the Lewis structure shown below.(a) Does the Lewis structure depict a neutral molecule or anion? If it is an ion, what is the charge on the ion? (b) What hybridizationis exhibited by each of the carbon atoms? (c) Arethere multiple equivalent resonance structures for the species?(d) How many electrons are in the p system of the species?arrow_forwardThe Lewis structure for the chlorate ion is :0: Calculate the formal charge on the chlorine (Cl ) atom. Express your answer as an integer. • View Available Hint(s) formal charge on Cl = Submit Part B Calculate the formal charge on each of the oxygen (O) atoms labeled a, b, and c in the following Lewis structure. :0: Express your answers as integers separated by commas. • View Available Hint(s) formal charge on Oa , Ob , Oc = Submit ormal charges to predict the most stable structure he interactive activity shows how to calculate the formal charge of atoms in a structure. These formal charges can be used to predict the resonance structure that contributes most to the stability of a molecule or ion. The struc enerally the most stable. Part C What are the formal charges on the sulfur (S), carbon (C ), and nitrogen (N ) atoms, respectively, in the resonance structure that contributes most to the stability of the thiocyanate ion, SCN¯ ? The possible resonance structures for the thiocyanate…arrow_forward(b) The Murchison meteorite that landed in Australia in 1969 contained 92 different amino acids, including 21 found in Earth organism A skeleton structure (single bond only) of one of these extraterrestrial amino acids is shown below. Draw a Lewis structure, and identify any atoms having a nonzero formal charge. H3N. C ČH2 ČH3 (c) Draw the orbital diagrams and Lewis symbols to depict the formation of Na* and CI ions from the atoms. Give the formula of the compound formed. (d) The predicted bond length for HF is 109 pm (the sum of the covalent radii of H, 37 pm and F, 72 pm), however the actual bond length for HF is shorter (92 pm). It was observed that the difference between predicted and actual bond lengths becomes smaller going down the halogen group from HF to HI Describe these observationsarrow_forward
- Help mearrow_forwardThe strength of a covalent bond depends upon the size of the atoms and the bond order. In general short bonds are strong bonds. For each pair of covalently bonded atoms, choose the one expected to have the higher bond energy. (A) CEO (B) C=O (C) C-N (D) CEN Use the References to access important values if needed for this question. (A,B) (C,D)arrow_forwardConsider the molecule QR3 , where Q has 7 valence electrons and R has 6 valence electrons. Both -1 elements are in the third period or below. (a) On your scratch paper, draw the BEST Lewis structure. Don't forget resonance, if applicable. (b) Show the formal charges for all elements.arrow_forward
- (a) Determine the formal charges on each atom and use them to determine the overall charge, if any, on the species represented by the following Lewis structure: :Ö:arrow_forwardThe cyanate ion is an anion consisting of one oxygen atom, one carbon atom, and onenitrogen atom, [OCN], in that order. a) Write three (3) resonance contributing structures for the cyanate ion. Show formalcharges.b) Based on your contributing structures, predict the O-C-N bond angle.arrow_forward1. Draw the Lewis structures for each of the following ions or molecules. For each, give (i) the molecular shape, (ii) the electron pair geometry at the central atom, and (iii) the hybridization of the central atom. (a) POF3 (b) XeO₂F3+ (c) BrCl₂ (d) N3 (the central atom is N; two other N's are bonded to it) (e) PF3arrow_forward
- What is the formal charge in this nitrogen atom?arrow_forward||| = 43°F Clear O ELECTRONIC STRUCTURE AND CHEMICAL BONDING Predicting the arrangement of electron groups around the centr... Answer the questions in the table below about the shape of the chlorine pentafluoride (CIF) molecule. How many electron groups are around the central chlorine atom? Note: one "electron group" means one lone pair, one single bond, one double bond, or one triple bond. What phrase best describes the arrangement of these electron groups around the central chlorine atom? (You may need to use the scrollbar to see all the choices.) Explanation Check 10 (choose one) (choose one) linear bent T-shaped trigonal planar trigonal pyramidal square planar square pyramidal tetrahedral sawhorse trigonal bipyramidal octahedral Q Search 0/3 2023 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center www.OTAPETY Accessibiarrow_forwardWrite resonance forms that describe the distribution of electrons in each of these molecules or ions. (a) sulfur dioxide, SO2 (b) carbonate ion, CO3²- (c) hydrogen carbonate ion, HCO3- (C is bonded to an OH group and two O atoms) (d) pyridine: H. H. (e) the allyl ion: H H H H-C-C-c-Harrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY