
College Physics
11th Edition
ISBN: 9781305952300
Author: Raymond A. Serway, Chris Vuille
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
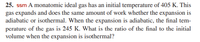
Transcribed Image Text:25. ssm A monatomic ideal gas has an initial temperature of 405 K. This
gas expands and does the same amount of work whether the expansion is
adiabatic or isothermal. When the expansion is adiabatic, the final tem-
perature of the gas is 245 K. What is the ratio of the final to the initial
volume when the expansion is isothermal?
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution
Trending nowThis is a popular solution!
Step by stepSolved in 2 steps

Knowledge Booster
Similar questions
- Question B: A sample of 1.00 mole of a diatomic ideal gas is initially at temperature 265 K and volume 0.200 m3. The gas first undergoes an isobaric expansion, such that its temperature increases by 110.0 K. It then undergoes an adiabatic expansion so that its final volume is 0,440 m3. i. Sketch a PV diagram for the two-step process, including labeled initial, final, and intermediate states, and a two-part curve/path with an arrow indicating direction. Label the initial state "i", the final state "f", and the intermediate state "b". Write down the known values for P, T, and V at each point, e.g. T; = 265 K, and Th = 375 K. (B.1) What is the initial pressure of the gas, Pi, in pascals [Pa]? Pi = Pa Enter a number. (B.2) What is the total heat transfer, Q, to the gas, in joules [J]? Q = Qtotal = (B.3) What is the total work done on the gas, w, in joules [J]? w = Wtotal = Enter your answer for problem (B.3) for credit. First, use the following questions as intermediate steps; answers can…arrow_forward3.0 moles of helium gas, that initially occupies a volume of 30L at a temperature of 280 K, isothermally expands to 40 L. How much work does the gas perform on its environment? а. 2.00 kcal b. 3.00 kcal с. 6.00 kcal d. 4.00 kcal е. 5.00 kcalarrow_forward4 di 8 Domanda 8 Completo Punteggio max.: 2 When an ideal gas is compressed isothermally Scegli un'alternativa: A. heat flows into the gas. OB. the internal energy of the gas does not change. OC. the work done on the gas is zero. OD. the temperature of the gas increases. Exam 17-12-2020: Revisione tentativo Domanda 9 O Cerca I https://elearning.unipd.it/dimed/mod/quiz/revien Barrow_forward
- You would like to raise the temperature of an ideal gas from 295 K to 960 K in an adiabatic process. a)What compression ratio will do the job for a monatomic gas? b)What compression ratio will do the job for a diatomic gas?arrow_forwardConsider ? = 5.00 mol of an ideal diatomic gas successively undergoing each one of the following thermodynamic processes:I. An adiabatic compression from an original volume of 0.150 m3to final volumeof 0.120 m3reaching a temperature of 293.0 K.II. An isothermal expansion to the original volume.III. An isochoric cooling to the original state. A) Draw a pressure-volume diagram showing all these processes.B) Calculate the pressure and temperature for each of the missing principal states.arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning University Physics (14th Edition)PhysicsISBN:9780133969290Author:Hugh D. Young, Roger A. FreedmanPublisher:PEARSON
University Physics (14th Edition)PhysicsISBN:9780133969290Author:Hugh D. Young, Roger A. FreedmanPublisher:PEARSON Introduction To Quantum MechanicsPhysicsISBN:9781107189638Author:Griffiths, David J., Schroeter, Darrell F.Publisher:Cambridge University Press
Introduction To Quantum MechanicsPhysicsISBN:9781107189638Author:Griffiths, David J., Schroeter, Darrell F.Publisher:Cambridge University Press Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Lecture- Tutorials for Introductory AstronomyPhysicsISBN:9780321820464Author:Edward E. Prather, Tim P. Slater, Jeff P. Adams, Gina BrissendenPublisher:Addison-Wesley
Lecture- Tutorials for Introductory AstronomyPhysicsISBN:9780321820464Author:Edward E. Prather, Tim P. Slater, Jeff P. Adams, Gina BrissendenPublisher:Addison-Wesley College Physics: A Strategic Approach (4th Editio...PhysicsISBN:9780134609034Author:Randall D. Knight (Professor Emeritus), Brian Jones, Stuart FieldPublisher:PEARSON
College Physics: A Strategic Approach (4th Editio...PhysicsISBN:9780134609034Author:Randall D. Knight (Professor Emeritus), Brian Jones, Stuart FieldPublisher:PEARSON

College Physics
Physics
ISBN:9781305952300
Author:Raymond A. Serway, Chris Vuille
Publisher:Cengage Learning

University Physics (14th Edition)
Physics
ISBN:9780133969290
Author:Hugh D. Young, Roger A. Freedman
Publisher:PEARSON

Introduction To Quantum Mechanics
Physics
ISBN:9781107189638
Author:Griffiths, David J., Schroeter, Darrell F.
Publisher:Cambridge University Press

Physics for Scientists and Engineers
Physics
ISBN:9781337553278
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning

Lecture- Tutorials for Introductory Astronomy
Physics
ISBN:9780321820464
Author:Edward E. Prather, Tim P. Slater, Jeff P. Adams, Gina Brissenden
Publisher:Addison-Wesley

College Physics: A Strategic Approach (4th Editio...
Physics
ISBN:9780134609034
Author:Randall D. Knight (Professor Emeritus), Brian Jones, Stuart Field
Publisher:PEARSON