
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
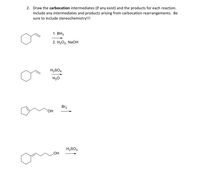
Transcribed Image Text:2. Draw the carbocation intermediates (if any exist) and the products for each reaction.
Include any intermediates and products arising from carbocation rearrangements. Be
sure to include stereochemistry!!!
1. ВНз
2. Н2О2, NaOн
H2SO4
H20
Br2
HO.
H2SO4
HOʻ
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution
Trending nowThis is a popular solution!
Step by stepSolved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- When this alkene is subjected to the conditions shown, two equivalents of a single product is formed. Draw the structure of the product that would be in the box to the right. 1. KMNO4, HO, H2O, heat -CH3 2. HCI H,C- Product 2 ČH3 ČH3arrow_forwardDraw the products of the following reactions, indicating both regiochemistry and stereochemistry when appropriate. Use wedge and hash bonds ONLY when needed to show reaction stereochemistry. In cases where there is more than one answer, just draw one.arrow_forwardProvide the structure of the product(s) with stereochemisty if appropriate. If multiple products indicate major product or if products are formed in equal amounts. If there is no reaction expected explain why. 1. H3180* 2. K2CO3 Mel CH3 1. CrO3, H30* H3C HO, 2. DCC, HN A, [-H2O] HOarrow_forward
- When the cleavage of an ether occurs, in step 2, how come the bromide ion does not attack the carbon on the cyclopentane? How come it attacks the CH2 carbon? Does it matter which one is being attacked?arrow_forward5. Which bromocyclohexane starting material would react faster - the cis or trans, and explain why? Draw both chair forms of the starting material to prove your point. Br K* OC(CH) K* OC(CH); Br cis transarrow_forwardDraw the reaction mechanism for the following transformation given the starting materials, product,and reagents. Be sure to include all intermediates,curvy reaction arrows,and include any formal charges.arrow_forward
- Draw the strcutres of the missing reactants, intermediates, or products in the following mechanisms. Include all lone pairs.arrow_forwardPredict the major organic product for each reaction indicate the correct stereochemistry of the product or applicable.arrow_forwardEach of these reaction produces ONE MAJOR PRODUCT. In each case, draw this product in the boxprovided, including appropriate stereochemistry when applicable. For multi-step reactions, just give theFINAL product, no intermediates.arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY