
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
thumb_up100%
hi i'm struggling a bit balancing this equation
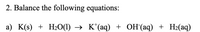
Transcribed Image Text:2. Balance the following equations:
a) K(s) + H2O(1)
K*(aq) + OH(aq) + H2(aq)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by stepSolved in 2 steps with 2 images

Knowledge Booster
Similar questions
- Options Decompostion rxn single replacement rxn double replacement rxn combustion rxnarrow_forward2. What coefficients would balance the following equation? _C2H6 + _02 –→_CO2+ _H2 O A. 1C2H6 + 502 → 2C02 + 3H20 B. 202H6 + 502→ 4CO2 + 6H20 С. 2С2H6 + 702 4C02 + 6H20 O D. 2C2H6 + 1002 → 4CO2 + 6H2Oarrow_forwardIndicate how many moles of the second reactant would be required to react exactly with 0.275 mole of the first reactantarrow_forward
- In a closed system, equal amounts of ammonia and oxygen react to produce nitrogen monoxide and water. What is the limiting reactant? NH3 4NH3 + 502 - 4NO + 6H,0 NO H20 OO Oarrow_forward100% Normal text Arial 11 B 1 - 1 2 3. 4 I5 6 7 8 T9 1 10 11 I 12 13 2. Copper (1) carbonate heptahydrate has the chemical formula a) CUCO3.5H20 b) CU2CO3.5H2O c) CuCoz. 7H20 d) Cu2CO3. 7H20 B. A double displacement reaction may be defined as: a) two or more simple substances producing a more complex substance *b) a more complex substance breaking down into simpler ones c) one element replacing another element in a compound d) a reaction involving two ionic compounds where the cations and anions replace each other Neutralisation occurs when: a) an acid is mixed with water b) a base is mixed with water c) a salt is mixed with an acid d) an acid is mixed with a base 山TOA Ai étv Book Proarrow_forwardH2O2(1) + SO2(g) -> H2SO4(aq) Express your answer as a chemical equation including phases. If the initial reaction is already balanced, enter the initial reaction.arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY