
Introduction to Chemical Engineering Thermodynamics
8th Edition
ISBN: 9781259696527
Author: J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark Swihart
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Question
For the debutanizer shown in Figure and considered estimate the Nmin by the Fenske equation. Assume a uniform operating pressure of 80 psia (552 kPa) throughout and utilize the ideal K-values given by Bachelor [4] as plotted in Figure.
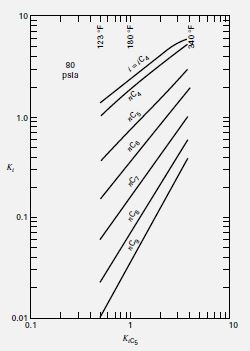
Transcribed Image Text:10
80
psla
1.0
к,
0.1
0.01
0.1
10
Kcs
ПT
1.
ПL
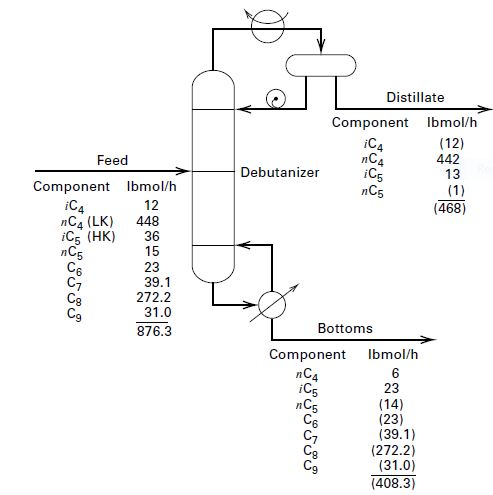
Transcribed Image Text:Distillate
Component Ibmol/h
iC4
пC4
iC5
пCs
(12)
442
13
Feed
Debutanizer
Component Ibmol/h
iC4
nCa (LK)
ic5 (HK)
(1)
(468)
12
448
36
15
23
39.1
272.2
31.0
Св
876.3
Bottoms
Component
Ibmol/h
6
23
nC4
iC5
nC5
C6
(14)
(23)
(39.1)
(272.2)
(31.0)
(408.3)
Cg
C9
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by stepSolved in 4 steps with 5 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemical-engineering and related others by exploring similar questions and additional content below.Similar questions
- A 3 meter tall aluminum tank with a 7 meter diameter is completely filled with ethanol. The empty weight of the tank is 250 kg. If the ethanol starts at 25 deg C, and there is 3000 kJ of heat available for transfer, what final temperature can be reached? And, what percentage of the heat transferred does the ethanol and aluminum each account for? (ethanol density = .789 kg/L)arrow_forward[1] Repeat Example 7.10 (pages 288-289) but with the following exceptions: water is discharged at 9,000 kPa, and n=0.8. Calculate the work of the pump and AT using steam tables and equation 7.25arrow_forwardplease show how to derive h using nusselt equation and solve for qheatlossarrow_forward
- i need the answer quicklyarrow_forwardchemical engineering A power plant emits 216 g of SO2 every hour. The wind recorded on the same day is 5 m s-1, and the atmospheric stability class is A for the clear summer afternoon. There is no plume rise, and the stack height is 30 m. The values of σy and σz are 215 m and 450 m, respectively. Determine: the concentration of SO2 0.7 km downwind along the plume centreline at ground level.arrow_forward2-37 The gage pressure in a liquid at a depth of 3 m is read to be 28 kPa. Determine the gage pressure in the same liquid at a depth of 9 m.arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 Introduction to Chemical Engineering Thermodynami...Chemical EngineeringISBN:9781259696527Author:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark SwihartPublisher:McGraw-Hill Education
Introduction to Chemical Engineering Thermodynami...Chemical EngineeringISBN:9781259696527Author:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark SwihartPublisher:McGraw-Hill Education Elementary Principles of Chemical Processes, Bind...Chemical EngineeringISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...Chemical EngineeringISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY Elements of Chemical Reaction Engineering (5th Ed...Chemical EngineeringISBN:9780133887518Author:H. Scott FoglerPublisher:Prentice Hall
Elements of Chemical Reaction Engineering (5th Ed...Chemical EngineeringISBN:9780133887518Author:H. Scott FoglerPublisher:Prentice Hall
 Industrial Plastics: Theory and ApplicationsChemical EngineeringISBN:9781285061238Author:Lokensgard, ErikPublisher:Delmar Cengage Learning
Industrial Plastics: Theory and ApplicationsChemical EngineeringISBN:9781285061238Author:Lokensgard, ErikPublisher:Delmar Cengage Learning Unit Operations of Chemical EngineeringChemical EngineeringISBN:9780072848236Author:Warren McCabe, Julian C. Smith, Peter HarriottPublisher:McGraw-Hill Companies, The
Unit Operations of Chemical EngineeringChemical EngineeringISBN:9780072848236Author:Warren McCabe, Julian C. Smith, Peter HarriottPublisher:McGraw-Hill Companies, The

Introduction to Chemical Engineering Thermodynami...
Chemical Engineering
ISBN:9781259696527
Author:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark Swihart
Publisher:McGraw-Hill Education

Elementary Principles of Chemical Processes, Bind...
Chemical Engineering
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY

Elements of Chemical Reaction Engineering (5th Ed...
Chemical Engineering
ISBN:9780133887518
Author:H. Scott Fogler
Publisher:Prentice Hall


Industrial Plastics: Theory and Applications
Chemical Engineering
ISBN:9781285061238
Author:Lokensgard, Erik
Publisher:Delmar Cengage Learning

Unit Operations of Chemical Engineering
Chemical Engineering
ISBN:9780072848236
Author:Warren McCabe, Julian C. Smith, Peter Harriott
Publisher:McGraw-Hill Companies, The