
Concept explainers
Which of the following structures are identical? (Green = Cl.)
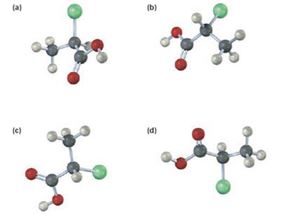
Interpretation:
Among the four structures given which are identical is to be stated.
Concept introduction:
All the four structures given have chiral carbons. By assigning the configuration as R or S the identical structures can be identified.
To state:
Which among the four structures given are identical.
Answer to Problem 26VC
Structures a, b, and d are identical. Structure c is different.
Explanation of Solution
The chiral carbon in all the four structures given is attached to the same four groups, H, Cl, CH3 and COOH but differently oriented. When the sequence rules are applied to these four groups, the order of priority obtained is Cl, COOH, CH3 and H. Using this priorities the configuration of the four structures can be obtained. It is clear that a, b and d have R configuration and d has S configuration. Hence a, b and d represent identical structures and c represents a different configuration.
Structure a, b, and d are identical. Structure c is different.
Want to see more full solutions like this?
Chapter 5 Solutions
Organic Chemistry
- 3. H2C=CH2 + KMNO, ?arrow_forward4. (a) Draw the structural formula for compounds D, E, G, H and J. H H2N- D HNO, CH3 G H,SO, A E J LIAIH, H,O*arrow_forwardThe initial reactive intermediate in the elimination reaction of 2-bromo-3-methyl-pentane is a tertiary carbanion terminal alkene primary carbon radical O tertiary alcohol O secondary carbocationarrow_forward
- Which pairs of structures are constitutional isomers? А B D ОН HO. HO. E F Harrow_forwardRank each of the representations of NOCl from best to worst, placing the best structure on top and the worst at the bottom.arrow_forward• -- H (b) H,C 3-methylhexane CH, (c) B கூ CH, CH, HgCG; சீகூ H₂CH H 3-ethyl-1-methyloctane 3,3 diethyl pentane 5Å, Å,CH, W CH3 CH2 CH₂ W21 Su2... CH3 X -CH3 G b F E L carrow_forward
- 8c) What is the relation between the following pairs H₂C O a. Cis-Trans O b. Structural O c. not isomers O d. Identical H3C CH3 H₂CH.arrow_forwardC5H8 has two elements of unsaturation (right?), which may indicate the presence of either double bonds or triple bonds or rings. Show all the possible structuresarrow_forwardDraw the structural formula of (3E,5E) 2,5-dibromo-3,5-octadiene. (3E,5E): draw the 3E,5E stereoisomer!!!arrow_forward
- H Br (CH3)3CO-K* H (CH)3СОНarrow_forward2b im confused ?arrow_forwardOrganic ChemistryFor each pair of compounds, indicate whether the first structure is lower, higher, or about the same in energy as the second structure. Be sure to explain. C angle (c) CH3 OH is OH in energy than/as H3C because: Po (d) CN CN NC- CN CO₂Et is in energy than/as CO₂Et CO₂Et EtO₂C because:arrow_forward
