
Concept explainers
Which compound in each pair is the stronger acid?
a.
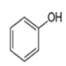 or
or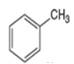
Trending nowThis is a popular solution!

Chapter 2 Solutions
Organic Chemistry (6th Edition)
- What is the relative trend in acidity and pKa of the two compounds? a. Structure I is the most acidic, and Structure I has the highest pKa. b. Structure I is the most acidic, and Structure I has the lowest pKa. c. Structure II is the most acidic, and Structure I has the highest pKa. d. Structure II is the most acidic, and Structure I has the lowest pKa.arrow_forwardIn each equilibrium, label the stronger acid, the stronger base, the weaker acid, and the weaker base. Also estimate the position of each equilibrium. (a) CH3CH2O + CH3CCH CH3CH2OH + CH3CC (b) CH3CH2O + HCl CH3CH2OH + Cl (c) CH3COOH + CH3CH2O CH3COO + CH3CH2OHarrow_forwardWhich compound in each pair is the stronger acid? c. CH3COOH or O,NCH,COOH a. CICH,COOH or FCH,COOH b. Cl,CHCH,OH or Cl,CHCH,CH,OHarrow_forward
- Which is a stronger base? a. HS− or HO− b. CH3O− or CH3NH c. CH3OH or CH3O d. Cl− or Br− e. CH3COO− or CF3COO− f. CH3CHClCOO− or CH3CHBrCOO−arrow_forward4. Rank the following compounds from strongest acid to weakest acid: ОН ОН TOH JOH I Y A B C Darrow_forwardWhich is the stronger base? a. Br- or I- b. CH3O- or CH3S-c. CH3CH2O− or CH3COO−d. H2C =CH e. FCH2CH2COO− or BrCH2CH2COO− f. ClCH2CH2O− or Cl2CHCH2O−arrow_forward
- Which of the following compounds is a strongest base? NH₂ O₂N 01 Oll O III ON OV NH₂ = II E III NH₂ IV NH₂ NH₂arrow_forwardWhat is the major product of this reaction? Ph. HO H,C H Ph H3C Ph Ph H3C Ph Ph Ph H H3C Ph Ph H2C H Pharrow_forward1. Rank each set of three alcohols in terms of increasing acidity, "1" being least acidic, and "3" being most acidic. goos CCI3 CI3C. Cl3 а. ОН ОН OH b. OH ОН ОН OH С. HOarrow_forward
 Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


