
Concept explainers
Draw short segments of the
- a. CH2 =CHF
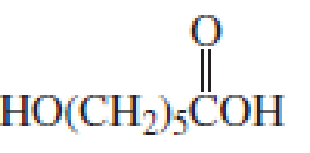
- b. CH2 =CHCO2H
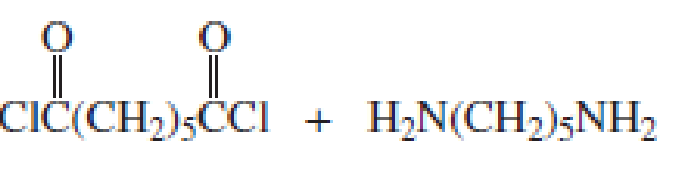
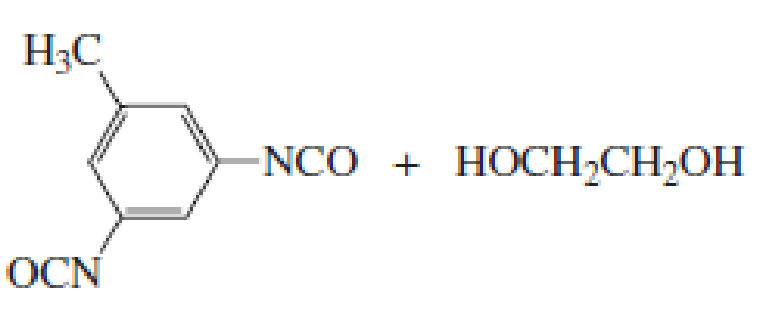
(a)
Interpretation:
The short segment of polymer obtained from the given monomer has to be drawn and also have to indicate whether it is a chain growth or step growth polymer.
Answer to Problem 22P
The short segment of given monomer is shown below and the monomer undergoes chain growth polymerization.
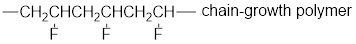
Explanation of Solution
Given the monomer is
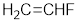
The short segment of polymer obtained from this monomer is given below.
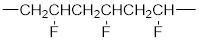
The polymer formed is a Chain growth polymer because monomer addition occurs at the end of the chain.
(b)
Interpretation:
The short segment of polymer obtained from the given monomer has to be drawn and also have to indicate whether it is a chain growth or step growth polymer.
Answer to Problem 22P
The short segment of given monomer is shown below and the monomer undergoes chain growth polymerization.
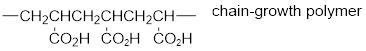
Explanation of Solution
Given the monomer is

The short segment of polymer obtained from this monomer is given below.
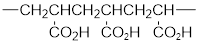
The polymer formed is a Chain growth polymer because monomer addition occurs at the end of the chain.
(c)
Interpretation:
The short segment of polymer obtained from the given monomer has to be drawn and also have to indicate whether it is a chain growth or step growth polymer.
Answer to Problem 22P
The short segment of given monomer is shown below and the monomer undergoes step growth polymerization.
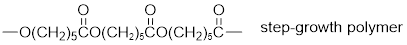
Explanation of Solution
Given the monomer is
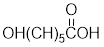
The short segment of polymer obtained from this monomer is given below.
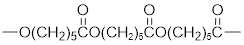
The polymer formed is a Step growth polymer because monomer addition occurs not at the end of the chain and these polymers are formed by combining monomers by removing small molecules of water or alcohol.
(d)
Interpretation:
The short segment of polymer obtained from the given monomer has to be drawn and also have to indicate whether it is a chain growth or step growth polymer.
Answer to Problem 22P
The short segment of given monomer is shown below and the monomer undergoes step growth polymerization.
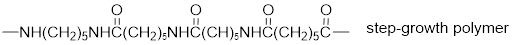
Explanation of Solution
Given the monomer is
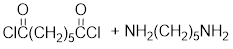
The short segment of polymer obtained from this monomer is given below.

The polymer formed is a step growth polymer because monomer addition occurs not at the end of the chain and these polymers are formed by combining monomers by removing small molecules of water or alcohol.
(e)
Interpretation:
The short segment of polymer obtained from the given monomer has to be drawn and also have to indicate whether it is a chain growth or step growth polymer.
Answer to Problem 22P
The short segment of given monomer is shown below and the monomer undergoes step growth polymerization.
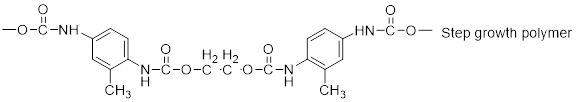
Explanation of Solution
Polymers are formed from linking small units called monomers and they are classified into Chain growth polymers and Step growth polymers.
Given the monomer is
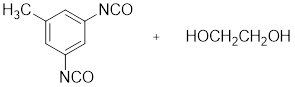
The short segment of polymer obtained from this monomer is given below.
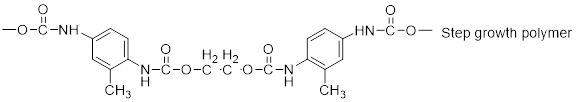
The polymer formed is a Step growth polymer because monomer addition occurs not at the end of the chain and these polymers are formed by combining monomers by removing small molecules of water or alcohol.
Want to see more full solutions like this?
Chapter 15 Solutions
Essential Organic Chemistry (3rd Edition)
- 1. Draw the structure of the monomer that is responsible for the following polymer: -CH,-CH-arrow_forwardDraw the structure(s) of the monomer(s) used to make each of the following polymers Define whether each polymer is a A. homopolymer or B. copolymer Define whether each polymer can be prepared by C. free radical polymerization or D. condensation polymerizationarrow_forward1. Draw the structure of the polymer formed by the following monomer. H H H-C=C- C = Narrow_forward
- Explain why styrene (CH2=CHPh) can be polymerized to polystyrene by all three methods of chain-growth polymerization.arrow_forwardAddition Polymerization: draw the polymer resulting from the addition polymerization of each of the following monomers: CH₂=CH₂ CH₂=CH CI CH₂=CH-CH3 CH₂=CH CF₂=CF₂ CH₂=CH N CH₂=C-CH₂ COCH, 0 CH₂=CH OCCH,arrow_forwardA thermosetting material called polyester is made when a carboxylic acid reacts with dialcohol. Write the condensation polymerization with byproduct. The monomers of carboxylic acid and dialcohol are respectively: HO-C -C-OH HO-CH,CH,-OHarrow_forward
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning





