
Concept explainers
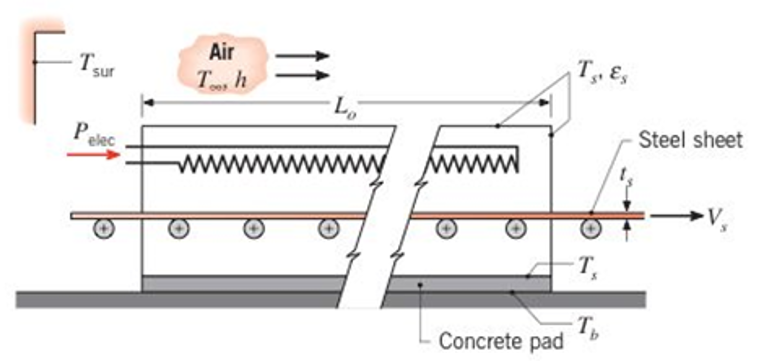
In one stage of an annealing process, 304 stainless steel sheet is taken from 300Â K to 1250Â K as it passes through an electrically heated oven at a speed of
Want to see the full answer?
Check out a sample textbook solution
Chapter 1 Solutions
Introduction to Heat Transfer
Additional Engineering Textbook Solutions
Thinking Like an Engineer: An Active Learning Approach (3rd Edition)
Applied Statics and Strength of Materials (6th Edition)
Mechanics of Materials
Engineering Mechanics: Dynamics (14th Edition)
Heating Ventilating and Air Conditioning: Analysis and Design
Degarmo's Materials And Processes In Manufacturing
- Estimate the volume of your kitchen oven and guess how many Watts it takes to pre-heat the air in your oven from 22 °C (72° F) to 221 °C (~430° F)? If the amount of steel that is heated in the inside of the oven totals 3 kg, how many Watts additionally does it take to pre-heat that? Time your oven to preheat and estimate the average Q needed.arrow_forwardExample 9: A 2-kW resistance heater wire whose thermal conductivity is k = 15 W/m ·°C has a diameter of D = 4 mm and a length of L = 0.5 m, and is used to boil water. If the outer surface temperature of the resistance wire is Ts = 105°C, determine the temperature at the center of the wire.arrow_forwardCalculate the heat capacity of a gas sample from the following information: The sam- ple comes to equilibrium in a flask at 25°C and 121.3 kPa. A stopcock is opened briefly, allowing the pressure to drop to 101.3 kPa. With the stopcock closed, the flask warms, returning to 25°C, and the pressure is measured as 104.0 kPa. Determine Cp in J mol- K- assuming the gas to be ideal and the expansion of the gas remaining in the flask to be reversible and adiabatic.arrow_forward
- Most automobiles have a coolant reservoir to catch radiator fluid that may overflow when the engine is hot. Such a radiator, made of copper, is filled to its 12-L capacity when at 10.0°C. -What volume of radiator fluid, in liters, will overflow when the radiator and fluid reach their 99.5°C operating temperature, given that the fluid’s thermal coefficient of volume expansion is 400.0 × 10-6 / °C? The coefficient of volume expansion for copper is 5.1 × 10-5 /°C.arrow_forwardFive hundred kilograms of poultry enter a chiller at 8°C and are frozen and chilled to a final temperature of 18°C for storage in 15 hours. The specific heat above and below freezing are 3.18 kJ/kg-°C and 1.55 kJ/kg -°C respectively. The latent heat is 246 kJ/kg and the freezing temperature is -5°C. Compute the product load.arrow_forwardK=519 A 1.5m diameter spherical balloon containing air at (K + 10) kPa is cooled such that the volume is reduced by 25%. For the process, the pressure is proportional to the square of the sphere’s diameter. What is the work done by the surrounding?arrow_forward
- Please show your complete solution. Thank you! A fluid of 700 kPa with a specific volume of 0.025m^3 and a velocity of 175 m/sec enters a device. Heat loss from the device by radiation is 23 kJ/ kg. The work done by the fluid is 465 kJ/ kg. The fluid exits at 136 kPa and 0.94 m^3/kg and 335 m/sec. What is the change of internal energy?arrow_forwardConsider two coffee cups A and B. Small coffee cup heaters are placed in cups A and B and heat is transferred to keep cup A at 45°C and cup B at 35°C. Room temperature is 25°C. The cups contain the same amount of water (100 grams). Which answer best describes the rates at which heat must be transferred to maintain the temperatures described above?: * A. Cup A will require heat at about five times the rate of B. B. Cup A will require heat at about twice the rate of B. C. Cup A will require heat at a slightly faster rate than B. D. Both cups will require heat at the same rate.arrow_forwardA gas with R = 278.6 J/kg-K and cv = .736 kJ/kg-K expands from 14.972 atm and 561 K to9.524 atm. Assume that throughout the process, the system is insulated. For 0.454 kg/sof this gas, prove that:a. V2 = 0.06 m3/s;b. dS = 0; andc. - dH = 1810 kJ/min.arrow_forward
- You have 500 g of water at 300C in a calorimeter with 20 cal / g heat capacity and a 400 g piece of aluminum is poured inside it at 900C. What is the equilibrium temperature of the system? Ignore losses to air. Specific heats: water: 1.00 cal / goC; aluminum: 0.21 cal / goC: the mass of the calorimeter is 20 cal/garrow_forwardA container of a gas is at 25°C and 270kPa. If it can withstand a pressure of 1000 kPa, at what temperature will it rupture?arrow_forwardThermodynamic problema Determine the final temperature of 0.8kg of glass with heat capacity of C = 0.092 cal/g°k and initial temperature of 40°C when heat of 580KJ is extracted.arrow_forward
 Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press
Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON
Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education
Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY
Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning
Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY
Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY





