When hydrochloric acid reacts with iron(III) oxide, water and iron(III) chloride are produced. The balanced equation for this reaction is: 6HCl(aq)+Fe2O3(s)------3H2O(1)+2FeCl3(aq) If 12 moles of hydrochloric acid react, The reaction consumes moles of iron(III) oxide. The reaction produces moles of water and moles of iron(III) chloride.
When hydrochloric acid reacts with iron(III) oxide, water and iron(III) chloride are produced. The balanced equation for this reaction is: 6HCl(aq)+Fe2O3(s)------3H2O(1)+2FeCl3(aq) If 12 moles of hydrochloric acid react, The reaction consumes moles of iron(III) oxide. The reaction produces moles of water and moles of iron(III) chloride.
Chemistry for Engineering Students
4th Edition
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Lawrence S. Brown, Tom Holme
Chapter4: Stoichiometry
Section: Chapter Questions
Problem 4.69PAE: 4.69 The pictures below show a molecular-scale view of a chemical reaction between H2 and CO to...
Related questions
Question
When hydrochloric acid reacts with iron(III) oxide, water and iron(III) chloride are produced. The balanced equation for this reaction is:
6HCl(aq)+Fe2O3(s)------3H2O(1)+2FeCl3(aq)
If 12 moles of hydrochloric acid react,
| The reaction consumes | moles of iron(III) oxide. | |
| The reaction produces | moles of water and | |
| moles of iron(III) chloride. |
Expert Solution
Step 1
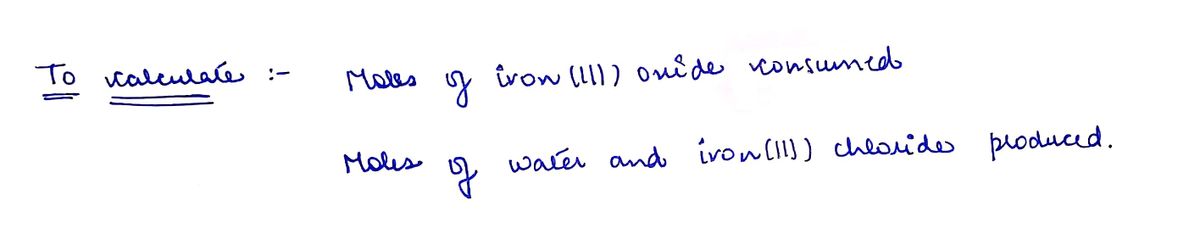
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781285199023
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781285199023
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781285199030
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

World of Chemistry
Chemistry
ISBN:
9780618562763
Author:
Steven S. Zumdahl
Publisher:
Houghton Mifflin College Div

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning