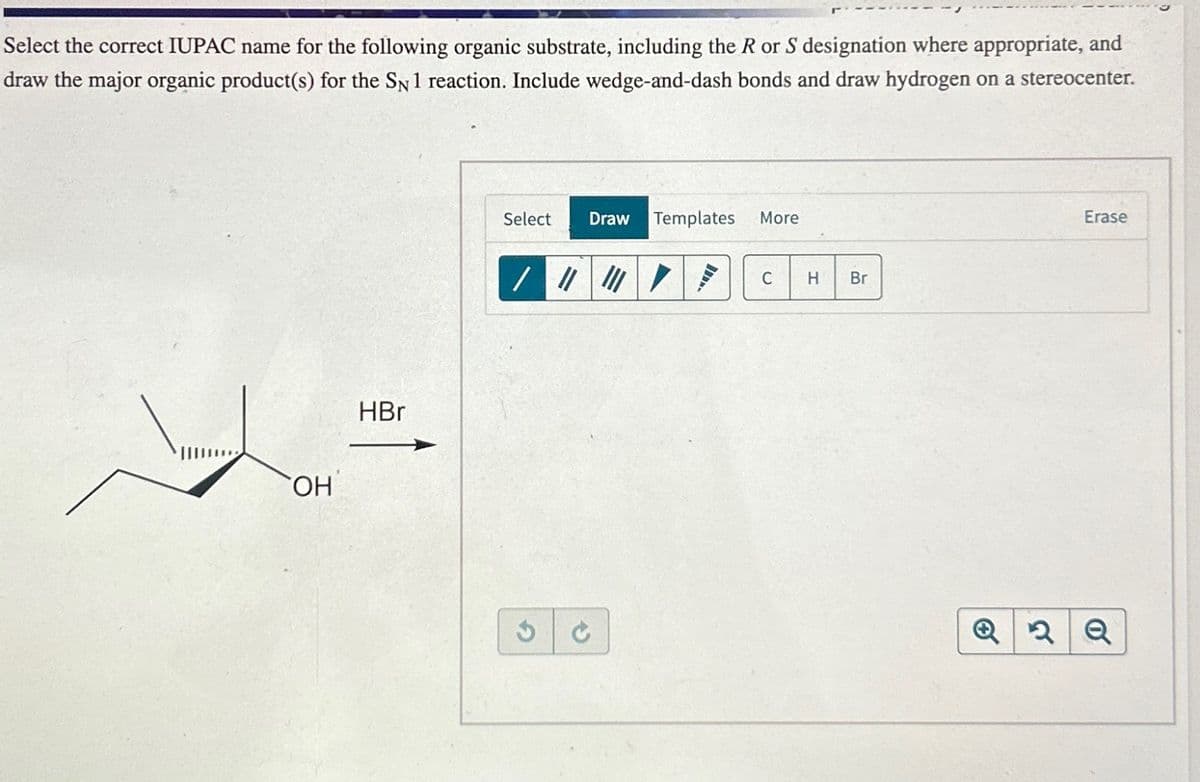
Select the correct IUPAC name for the following organic substrate, including the R or S designation where appropriate, and draw the major organic product(s) for the SN 1 reaction. Include wedge-and-dash bonds and draw hydrogen on a stereocenter. OH HBr Select Draw Templates More G . C H Br Erase Q2Q
Q: Draw Zaitsev and Hofmann products that are expected when each of the following compounds is treated…
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: If you know the starting mass of the limiting reactant in a balanced equation, you can only…
A: The objective of the question is to determine whether it is possible to calculate the mass of…
Q: Predict the major products of the following organic reaction:
A: The Diels–Alder reaction is a pericyclic reaction (cycloaddition) between a conjugated diene and a…
Q: Draw a structural formula for the major product of the reaction shown. CHa CHICHCH=CH₂ Cl₂ H₂O Show…
A:
Q: Determine [OH−] of a solution that is 0.180 M in F−. (Ka for HF is 6.8×10−4)
A: 1.6 * 10^ -6 MExplanation:use:Kb = Kw/KaKw is dissociation constant of water whose value is…
Q: What is the pH of a solution of 0.300 M HNO containing 0.110 M NaNO? (Ka of HNO is 4.5 \times 10)
A:
Q: Chemistry 421 C-13 Matching Homework: Write the letter of the structure on the spectrum that it…
A: Given are 13C NMR spectra for organic compounds.In 13C NMR, number of signals is equal to number of…
Q: Calculate a value for the equilibrium constant for the reaction O2(g) + O (g) = O3(g) given…
A: The objective of the question is to find the equilibrium constant for the reaction O2(g) + O (g) =…
Q: E. Calculating thermodynamic quantities (Individual Work) Temperature (°C) Temperature Ksp (K) AG°…
A: The equilibrium constant, temperature, enthalpy, and entropy are related according to the following…
Q: If 36.7 g of AgNO₃ react with 28.6 g of H₂SO₄ according to this UNBALANCED equation below, how many…
A: The objective of this question is to determine the amount of Ag2SO4 that could be formed from the…
Q: 1 per hift tab caps lack Check the box under each aldohexose. If there are none, check the None of…
A: All answers are uploaded Explanation:
Q: Show the retrosynthetic analysis and forward synthesis of the following compound. 7. H3C c -COOH
A: The objective of this question is to write the retrosynthetic pathway and forward reaction of the…
Q: Provide a synthesis for the target molecule shown below, starting with compounds that contain no…
A:
Q: Draw the major product of the following reaction. CH3 HBr peroxides
A: In the given reaction the starting compound is an alkene and the reagent is..The given reaction is…
Q: Problem 13 of 14 Submit A common ketone starting material is shown below. Predict the major product…
A: Answer shown in attachment provide in explanation box Explanation:
Q: The equilibrium constant, Kc, for the following reaction is 1.29×10-2 at 600 K.COCl2(g) CO(g) +…
A: The objective of the question is to calculate the equilibrium concentrations of reactant and…
Q: Fill in the boxes with the product of the following reactions of alkenes. Draw only the PREDOMINANT…
A: The objective of this question is to draw the products for the given reactions.
Q: A researcher has synthesized an oligopeptide "SLISRAEKLAGVRVGV" and needs to confirm whether the…
A: Introduction:Peptide synthesis is a fundamental technique in biochemistry and molecular biology,…
Q: Which of the following is an SN1 reaction? HO H₂O (S) (R) O b. NaOH OH + NaⱭ H₂O Ос HO NaOH + NaCl…
A: SN1: Racemization or retention of configuration is possible due to the formation of a planar…
Q: (please show reaekson and incorrect option explain)
A: The objective of the question is to identify the product formed when CH2CH2CH2-OH reacts with HBr.
Q: Draw the major product of this reaction. Use a dash or wedge bond to indicate stereochemistry of…
A: In this reaction alkyl bromide has been turned into amine.
Q: Draw the appropriate Newman projection which leads to the major E2 ELIMINATION product. Hint: Review…
A:
Q: Show the starting diene and dienophile you could use to prepare the following molecule: Diene +…
A: Given,The reaction is:
Q: what is the name of the structure OH о N
A: IUPAC is an acronym for the International Union of Pure and Applied Chemistry. It is an…
Q: The compound below has multiple electrophilic sites which could potentially react with an…
A: Given reaction:The given compound has multiple electrophilic sites which could potentially react…
Q: (A) the rate of substitution doesn't depend on nucleophile concentration and (B) the product…
A: The question aims to find out whether the given statements are correct for the given substrates and…
Q: A student ran the following reaction in the laboratory at 703 K: H2(g) + I2 (g) = 2HI (g) When…
A: The objective of the question is to calculate the equilibrium constant, Kc, for the reaction H2(g) +…
Q: Why do we use dilute HCl for the first step and concentrated HCl for the second step? о OH NaBH HCI…
A: Secondary alcohols can undergo SN2 and SN1 reactions to form alkyl halide:Dilute HCl (aqueous): In…
Q: Make the following changes on the molecule in the drawing area below: 1. First, make sure this is…
A: α (Alpha) – the name given to the configuration of a cyclic sugar where the oxygen on the anomeric…
Q: Question 9 Draw major E2 ELIMINATION product. Hint: Review Chapter 10.3, the elimination can only…
A: This reaction is an example of E2 reaction in which final product is always an alkene
Q: ) Find out the four quantum numbers of the 17 th electron of the element Ar Ans: provide a solution…
A: We are given argon (Ar) We have to determine the quantum numbers of the 17th electron.
Q: 1) Explain why the protonation of pyrrole occurs at C2 to form A, rather than the N atom to form B…
A: pKa = 0.4pKa =5.3
Q: Draw a structural formula for the more stable carbocation intermediate formed H3C C=CH2 + HBr H3C •…
A: Answer with explanation is provided in the image. Explanation:
Q: Fill in the boxes with the product of the following reactions of alkenes. Draw only the PREDOMINANT…
A: Alkene reacts with hydrohaloacid or H2O/ H2SO4 gives gives alkyl halide and alcohol respectively.
Q: Q write mechansim Two reactions: (1) 1., H (1 equiv.) 2. a) LiAlH b) H2O work - up 3. H (final…
A: The objective of the question is to write the mechanism for the given reactions. However, the…
Q: Calculate the percent ionization of a 0.20 M benzoic acid solution in a solution containing 0.25…
A: The objective of this question is to calculate the percent ionization of a 0.20 M benzoic acid…
Q: What is the pH of a 4.1 × 106 M KOH solution?
A: The objective of this question is to calculate the pH of a solution of potassium hydroxide (KOH).
Q: When the following equation is balanced properly under acidic conditions, what are the coefficients…
A: The objective of the question is to balance the given redox reaction under acidic conditions and…
Q: Show the retrosynthetic analysis and forward synthesis of the following compounds. ง c 2. OCH3
A: The question is based on the concept of organic synthesis.We need to synthesize the product using…
Q: Phthalonitrile (C2H4N2) is produced by the ammoxidation of oxylene (C 8H10) according to the…
A: The balanced equation for givenreaction is C8H10(l) + 3O2(g) + 2NH3 (g) → C8H4N2 (s) + 6H2O(l)moles…
Q: H₂C H₂C Write the products of your compound and the following reagents. If the reaction would not…
A: Aldehydes react with to get oxidized to carboxylic acids.
Q: A positive anti-RNP in the antinuclear antibody group is suggestive of which autoimmune disease?…
A: The objective of the question is to identify the autoimmune disease that is suggested by a positive…
Q: 1. Write a mechanism for the formation of oximes from aldehydes and hydroxylamine. 2. Reaction of…
A: The objective of the first part of the question is to describe the mechanism for the formation of…
Q: Draw the structure of 4-ethoxy-2,2-dimethyldecane.
A: Compound 4-ethoxy-2,2-dimethyldecane is given.Draw the structure of this compound.The structural…
Q: Please describe the relationship between the compounds in each pair? Type [with exact spelling] one…
A: Identical: Same moleculeEnantiomers: Optical isomers which are non-superimposible mirror images of…
Q: Suppose a 250. mL flask is filled with 0.90 mol of Cl₂, 1.2 mol of HCl and 2.0 mol of CC14. The…
A:
Q: What is the IUPAC for the following compound?
A: Organic compounds can be defined as compounds that contain carbon and hydrogen atoms. We have been…
Q: explain how mass spectrometer operates describing how particles are selected
A: This question aims to explain the work of a mass spectrometer and describe how the particles are…
Q: The equilibrium constant, Kp , for the following reaction is 1.04×10-2 at 548 K.NH4Cl(s) NH3(g) +…
A: The objective of the question is to find the partial pressure of HCl(g) in the equilibrium mixture.
Q: Show the retrosynthetic analysis and forward synthesis of the following compounds. a. b. OH -OH CHO
A: The question is based on the concept of organic synthesis.We need to synthesize the product using…
V
Step by step
Solved in 3 steps with 2 images

- Qa 18. Bromination of 5a-cholestan-3-one occurs at C-2 or C-4 to give 2 regioisomers. Two unsaturated ketones with lambdamax = 230 nm and lambdamax = 241 nm are yielded during dehydrobromination. Use Woodward-Fieser rules to distinguish between the two.Which of the following Phase 2 reactions is likely to target a N-bearing functional group specifically? a Acetylation b Glycine conjugation c Glucuronic acid conjugation d SulfationGive the name/s only (structures not required) of the product for the reaction of (i) A and (ii) B with the reagents listed below. Your answers should be given using A1 -A10 and B1-B10 as indicators.Eg. the product for your first reaction should be stated as A1 = name of product etc. b2h6/h202 n-bromosuccinimide kmno4/oh-
- PLEASE HELP Analyze and compare the spectra of the reactants and products in this Esterification chemical reaction. label the spectra, identify differences between them, and discuss how these differences support the formation of Isoamyl acetate. PLEASE BE SPECIFIC TO THIS SPECTRA POSTED, NO GENERAL RESPONSE. esterification experiment: Acetic acid + Isoamyl alcohol = Isoamyl acetate + H20thanks. multiple choice: 1. Which of the following is true regarding an SN1 reaction? a. It would be faster at 25 ̊ than 50 ̊ b. It would be faster in ethanol than in pentane c. Keeping the moles of reactants constant but doubling the quantity of solvent would decrease the rate by a factor of 4. d. Stereochemical inversion occurs exclusively 2. Which of the following statements is true? a. The rate determining step is always the last step in a reaction mechanism. b. The stability/reactivity principle says that the more stable of two chemicals will be more reactive c. The reactivity/selectivity principle says that the more reactive of two chemicals will be less selective. d. The activation barrier for a reaction is the difference in energy between reactants and final products. 3. Which of the following statements is FALSE? a. Optically active solutions always contain chiral molecules. b. Two diastereomers always have identical melting points c. Optically…Please answer ASAP Draw the arrow-pushing mechanism for the following reaction. 1) P(OCH2CH3)3, NaH 2) 4-Methyl-2-Pentanone
- For each of the substrates below, identify whether: (A) the rate of substitution doesn't depend on nucleophile concentration and (B) the product distribution from substitution gives a 50/50 mix of enantiomers. If you answered "yes" for the first susbtrate, draw the intermediate that forms during a nucleophilic substitution reaction in the space below the table.For each of the substrates below, identify whether: (A) the rate of substitution doesn't depend on nucleophile concentration and (B) the product distribution from substitution gives a 50/50 mix of enantiomers. If you answered "yes" for the first substrate, draw the intermediate that forms during a nucleophilic substitution reaction in the space below the tableI recovered 3 mL of cyclohexene, how many grams of water was produced in the reaction please show how? Cyclohexanol can attack the carbocation intermediate formed during the synthesis of cyclohexene. Draw this side product with the molecular formula C12H22O. Please ASAPPPP you guys didn't answer the last one
- The E1 mechanism (unimolecular elimination) of Elimination ?Draw a mechanism reaction from week 1 and 2 A wittig reaction experiment: Week 1 -》 preparation of phosphonium salt Materials : triphenylphosphine (5.3g),methyl Bromoacetate 3.36g (2.1ml) , ethanol 30ml . The yield obtained was 8.93g .The percentage yield was 106.4% Week 2 -》 Formation of the ylide and wittig reaction Materials :Phosphonium salt (5g), napthyl-2- carboxaldehyde( 2.65g), 20 ml of water and 5 nl of Nahco3. Yield obtained was 0.51g . The percentage yield was 14.2%. Week 3: Solvent free wittig reaction Materials: Benzyltriphenylphosphonium chloride (0.5g ), 4 - Bromoabenzaldehyde (0.24g), Potassium phosphate (tribasi c) 0.275g .The yield obtained was 1.21g. The percentage yield was 35.6%.Why are SN1 reactions favored by polar protic solvents? Group of answer choices Polar protic solvents stabilize reaction intermediates Polar protic solvents cannot form hydrogen bonds. Polar protic solvents contribute leaving groups. Polar protic solvents contribute leaving groups.

