Q: Provide the correct product(s) for the following reaction
A: This reaction is a cross aldol reaction.
Q: NH2
A: The given reagent is Di-tert-butyl dicarbonate which is commonly known as Boc anhydride. It is a…
Q: 2. Propose a full mechanism using curvy reactions arrows for the following transformation. A
A: See the below for curvy mechanism of given transformation
Q: Propose a plausible mechanism for the following reaction: (H,SOJ
A: Interpretation - To tell about the possible reaction mechanism for the following given reaction…
Q: b) Qax HO
A:
Q: b) Propose a detailed reaction mechanism for the following conversion. он Acetone HCI но но. -OH OH…
A: Acetone /HCl is used for the protection of syn-1,2-diol. For protection with acetone/HCl glucose…
Q: When the illustrated reagents are treated with a strong base the indicated reaction is observed to…
A: Witting reagent contains triphenyl as substituents and utilized in the generation of alkene. In the…
Q: (d) Propose a reaction mechanism to account for the following reaction. AICI3
A: In this question we have to tell the mechanism of the given reaction.
Q: Indicate the directivity of the molecule below, and indicate if it is an 'activator' or a…
A: NO2(nitro group) is considered as an electron withdrawing group(EWG)
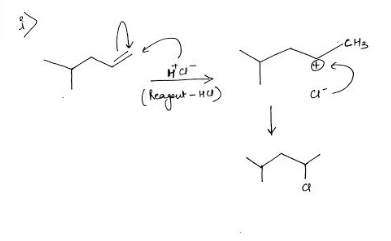
Q: Propose
A: When alcohol is heated then water molecule gets eliminate and we get alkene.
Q: 1. Provide a detailed mechanism for the following reaction and give the product. H2SO4
A: First c=O will attack on H+ and carbonyl carbon will protonate and then H2O will attack and…
Q: MgBr O CI 2) H30* pyridine
A: See mechanism below.
Q: 2. Propose a synthesis for the following compound starting with benzene. NO, CI
A:
Q: a. Propose a synthesis for compound D shown below, starting with benzene. Identify appropriate…
A: Here we have to propose a synthesis of the given compound from starting material from benzene in…
Q: 1.Provide the identity of Intermediate I and Intermediate Il in the following synthetic sequence,…
A:
Q: Which of (a)-(d) shows the increasing order of basicity of compounds 1-4?
A:
Q: Minor product via E1 for 2-butanol and HCl?
A: Alcohol reacts with hydrogen chloride to give substitution and elimination reactions, depending on…
Q: c) Outline a synthesis for compound G shown below starting with a 2- hydroxybenzylketone.
A:
Q: Represent the structures of intermediaries (A – C) and product D. Propose a mechanism for each of…
A: A molecular entity known as a reaction intermediate or an intermediate is created from the reactants…
Q: Propose a mechanism for this isomerization. ARSO3H
A: Any molecule or ion which are electron-rich species is called nucleophile, for example -NH2,-OH, and…
Q: 2. Provide the product for the following reaction. Provide the sterochemistry an regiochemistry…
A: Hello. Since the question contains multiple subparts the first three subparts are solved. In case…
Q: provide the mechanism and reagents to perform the reaction. Br Но
A: To provide the mechanism and reagents for the given reaction
Q: 3. Propose a reasonable synthesis for the following transformation. H
A:
Q: aq Br2
A:
Q: With reference to the following reaction scheme: LIAIH4 Et,NH HCI (cat.) A C3H180 B C12H25N Me Но…
A: We have to draw the structures of the compounds A-D.
Q: 2. Provide a detailed mechanism for the following reaction: NaOH + NH3 `NH, H20
A: When NH2 group react with H form a good leaving group NH3
Q: CH3 CH=CH, H*
A: The given reaction, When 2 molecules of styrene is treated with aqueous sulfuric acid,…
Q: Propose a detailed mechanism for the reaction below. لعلام , AICI3
A:
Q: Suggest efficient synthesis for the following transformations. `Br (a) он (b) ICH3
A:
Q: For the compound below, propose an efficient synthesis using diethyl as a starting material. Provide…
A:
Q: 2. Propose a reasonable synthesis of compound 2 from compound 1. Use no more than 4 steps. Он он он…
A: "Ardnt-Eistert Homologation" is mainly used for the conversion of a carboxylic acid to larger…
Q: Propose a synthesis for the formation of compound A below. Start with benzene and inorganic reagents…
A: Here we have to propose a synthesis for the formation given compound starting from benzene in…
Q: H20, H2SO4 -OH-
A:
Q: Provide the major organic product for the following reaction. iH H₂NOCH3, H
A: “Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: 3. Provide full mechanism (curved arrows and intermediates) for the following reaction. OCH3 1.…
A: Answer 3 ) The mechanism for the given reaction = ? Note : As per our company guidelines, we are…
Q: H2SO4 H2O
A: Alkene on reaction with water in acidic medium gives the corresponding alcohol. The first step is…
Q: Ph H₂SO4 Ph SO₂H Producto minoritario SO₂H Producto mayoritario
A:
Q: the following reaction go through an SNl or Sn2 mechanism, both, or not proceed as written? Explain…
A:
Q: Q2. Provide reasonable reagents and a reasonable mechanism to facilitate the transformation shown in…
A:
Q: II. Propose a suitable synthetic route to carry out the following transformations.
A:
Q: Complete the Reaction below and provide the following (i) a detailed stepwise mechanism to account…
A:
Q: OH H2SO4 H20
A:
Q: 2) Provide the reagent(s) and the reaction mechanism of the following reaction. (* is the radio…
A: Detail mechanistic pathway is given below to find out the correct reagents for following conversion
Q: Provide the appropriate reagent(s) and conditions for the following transforma ベロで NH2 Ph Ph Ph Ph.
A:
Q: Choose the best reagent (A-E) for carrying out the following conversion from the list provided…
A: Given : structure of reactant and products. Here oxidation of alcohol is happening.
Q: (b) Suggest a synthesis of each of the following compounds, starting from suitable precursors.…
A: Carbene react with alkene to give corresponding cylcoproane derivatives. Reaction of carbene is…
Q: 1.A. Complete the following reaction and provide a detailed, step-by-step mechanism for the process.…
A:
Q: ОН HO СООН C-OCH3
A: There is the conversion of o-hydroxybenzoic acid to o-hydroxyacetophenone.
Q: Proposing a plausible arrow pushing mechanism for the following transformation. Show all steps. H3O*…
A: In the above reaction, the double bonded O of the ester group will take proton first. Now the other…

Step by step
Solved in 2 steps with 2 images

- 1) Provide the reagent(s) and the reaction mechanism of the following reaction. H₂N#16b. Provide the missing reactants, reagents, or products for the following reaction sequences below.4. identify the structure of A and B in the following synthetic scheme: Write out complete reactions for each step, showing the structure of all reactants and products. (a) cyclohexanol +Na2Cr2O7/H2SO4, H2O -------> A (b) A + Et2NH/H2SO4 ----->B
- Provide the major product for the following reaction? (1) BH3, ether (2) H2O2, OHi) ii) 1. Propose a mechanism for the following reactions. H (CH3)2NH H* NaOH EtOHChemistry 3. Provide reagents/conditions to accomplish the following syntheses. Several steps are required in some cases. (c) (d) H2N. NH2 но H. NH2 но
- Determine whether each of the following syntheses requires a reaction that alters the carbon skeleton. (a) (b) OH (c) (d) OCH,CH3 CH3 (e) OH (f) H,CO он H;CO. (g) H3CO, (h) H3CO. NO2 NO2Provide the reagents necessary to complete the following reactions. More than one step may be necessary, if so number separate steps.b) Refer to the following equation to answer Q3b (i), (ii) and (iii). CH3 H,SO, Н—с—он C-CH3 ? + H2O Но- ČH3 (i) Determine the product of the above reaction. (ii) Name the above reaction. (iii) Propose the mechanism for the above reaction.
- a) Provide a mechanism that accounts for the product formed in the following reaction. OH CH H" ► 1-ethoxy-1-methyleyclohexane + CH,CH,OH b) Show the structure of the product(s) formed in the following reaction and write the mechanism for the reaction. OH CH + (CH;);COHGive the major product(s) of the following reaction. 1) LIAIH, 2) H;0*Provide the structure(s) of the expected major organic product of the reaction shown. 1) Disiamylborane 2) H₂O₂, NaOH OI O II ||| O IV OV CH3CH₂C(CH3)2C=CH OH OH xx xo IV

