One way the U.S. Environmental Protection Agency (EPA) tests for chloride contaminants in water is by titrating a sample of silver nitrate solution. Any chloride anions in solution will combine with the silver cations to produce bright white silver chloride precipitate. Suppose an EPA chemist tests a 200. mL sample of groundwater known to be contaminated with tin(II) chloride, which would react with silver nitrate solution like this: SnCl,(aq) + 2 AgNO;(aq) → 2 AgCl(5) + Sn(NO3),(aq) The chemist adds 23.0 mM silver nitrate solution to the sample until silver chloride stops forming. He then washes, dries, and weighs the precipitate. He finds he has collected 6.8 mg of silver chloride. Calculate the concentration of tin(II) chloride contaminant in the original groundwater sample. Be sure your answer has the correct number of significant digits.
One way the U.S. Environmental Protection Agency (EPA) tests for chloride contaminants in water is by titrating a sample of silver nitrate solution. Any chloride anions in solution will combine with the silver cations to produce bright white silver chloride precipitate. Suppose an EPA chemist tests a 200. mL sample of groundwater known to be contaminated with tin(II) chloride, which would react with silver nitrate solution like this: SnCl,(aq) + 2 AgNO;(aq) → 2 AgCl(5) + Sn(NO3),(aq) The chemist adds 23.0 mM silver nitrate solution to the sample until silver chloride stops forming. He then washes, dries, and weighs the precipitate. He finds he has collected 6.8 mg of silver chloride. Calculate the concentration of tin(II) chloride contaminant in the original groundwater sample. Be sure your answer has the correct number of significant digits.
Chapter2: Basic Statistical Analysis With Excel
Section: Chapter Questions
Problem 12P
Related questions
Question

Transcribed Image Text:因
+ II
P
8
团
w/
S - Cameron Hite Learn
b Chemistry Questions & Answers
+
A www-awu.aleks.com/alekscgi/x/Isl.exe/1o_u-IgNslkr7j8P3jH-IVDWKVW_BBZZI6tTytly4Fcfu6zOtOf8oMM9sLjOBhHfJ6ZNhysAi_9KCqM8FRGCtiPxvdUuxrTTETxcEo9QVTr9NhEkLqSLo?1oBw7QYjlbav. E
O CHEMICAL REACTIONS
0/3
Cameron V
Solving for a reactant in solution
One way the U.S. Environmental Protection Agency (EPA) tests for chloride contaminants in water is by titrating a sample of silver nitrate solution. Any chloride
anions in solution will combine with the silver cations to produce bright white silver chloride precipitate.
Suppose an EPA chemist tests a 200. mL sample of groundwater known to be contaminated with tin(II) chloride, which would react with silver nitrate solution
like this:
SnCl,(aq) + 2 AgNO,(aq) → 2 AgCl(s) +
Sn(NO,),(aq)
The chemist adds 23.0 mM silver nitrate solution to the sample until silver chloride stops forming. He then washes, dries, and weighs the precipitate. He finds he
has collected
6.8
of silver chloride.
Calculate the concentration of tin(II) chloride contaminant in the original groundwater sample. Be sure your answer has the correct number of significant digits.
Explanation
Check
O 2022 McGraw Hill LLC. All Rights Reserved. Terms of Use I Privacy Center Accessibility
4:19
2/26/20
-D
+D
F5
to
PrtSc
F7
F10
F11
F12
Insert
Delete
%23
&
2
%24
3.
Backspace
4.
9.
7.
R.
A.
Expert Solution
Step 1
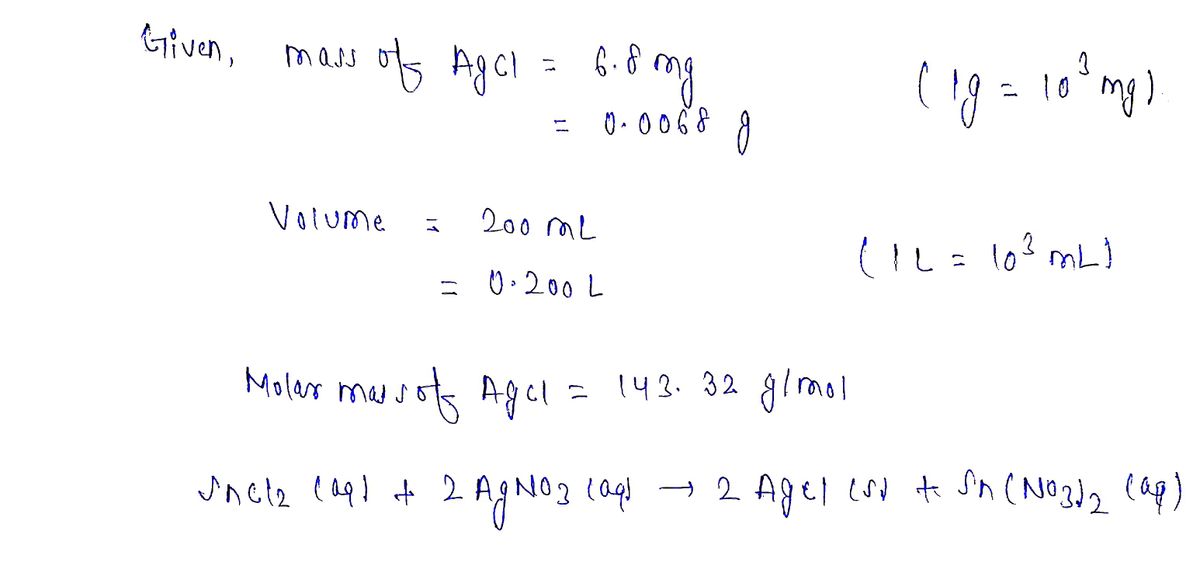
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
Recommended textbooks for you

