Q: Consider the following reaction: 2Mg(s) + O2(g) →2MgO(s), AH=-1204 kJ endothermic Submit Previous…
A:
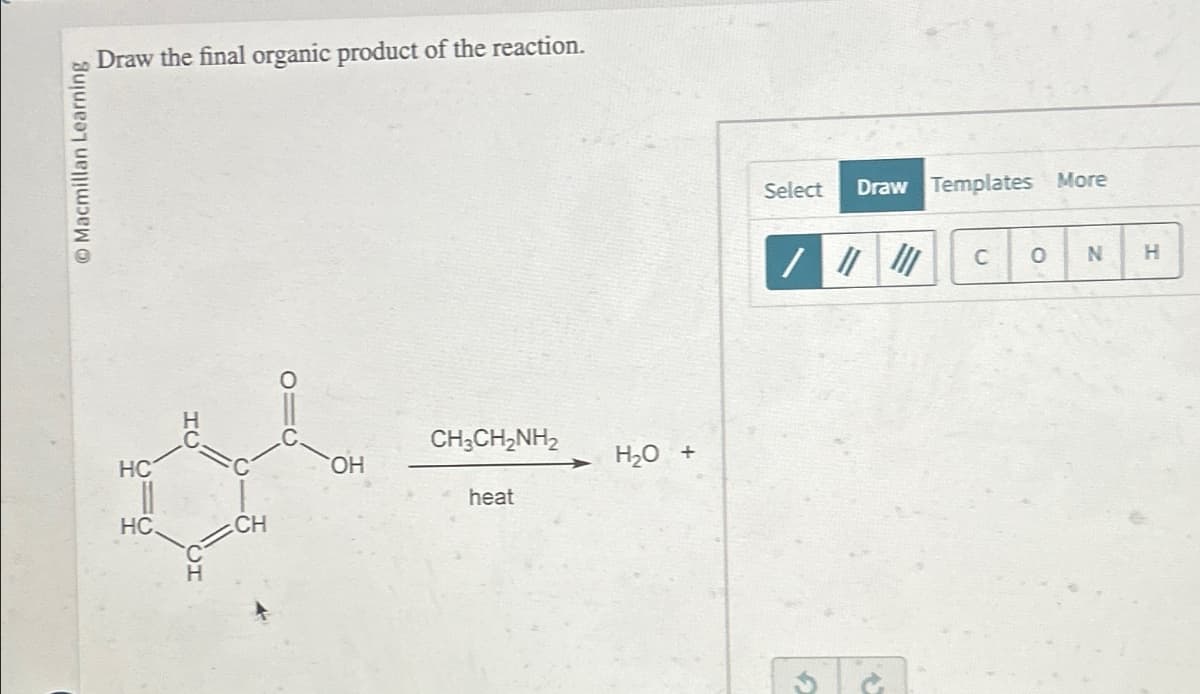
Q: 7) Predict the major product of the reaction. CH3
A: Organic reactions can be defined as the reactions in which organic reactants react with each other…
Q: At 22 °C, an excess amount of a generic metal hydroxide, M(OH),, is mixed with pure water. The…
A: Temperature = of the salt at = ?
Q: With reference to the H-NMR spectra given below, answer the following: For the starting material,…
A:
Q: Draw major E2 ELIMINATION product. Hint 1: Review Chapter 10.3, the elimination can only occur if…
A: For an E2 elimination to take place leaving group (Br in this case) and hydrogen at beta carbon (H…
Q: 3. Stereochemistry. Give the structure of the major product formed for each of the following…
A:
Q: After the formation of the enolate anion, the intramolecular nucleophilic addition takes place.
A: Answer shown in attachment provide in explanation box Explanation:
Q: 4 Devise a synthesis of each compound from cyclohexene as the startin material. More than one step…
A: Cyclo alkane to given compounds prepared by below methods.
Q: please show the correct arrows that will create the product Mg ^ Select to Edit Arrows H3O+ H' H H…
A: Information about the question
Q: Consider the reaction below. Then select all that are true about this reaction. CH,CH₂ÖH H CH,CH,OH…
A: Answer:In any reaction, specie that loses its H+ ion is called Bronsted-Lowry acid and specie that…
Q: Fill in the missing chemical formulae in the tables below. acid conjugate base base conjugate acid…
A: Conjugate pair of acid and base:A conjugate pair of acid and base differs by a proton only.
Q: For each of the following acid-base reactions, calculate how many grams of each acid are necessary…
A: The objective of the question is to calculate the amount of acid (HNO3) in grams required to…
Q: Calculate the cell potential for the galvanic cell in which the given reaction occurs at 25 °C,…
A: In the given question->[Sn2+] = 0.0670 M[Co3+] = 0.0365 M[Sn4+] = 0.00661 M [Co2+] = 0.00991MThe…
Q: 1 2 3 or or 4
A: Two esters compound react in the presence of a strong base. The reaction produces a beta-keto ester
Q: Draw the appropriate Newman projection which leads to the major E2 ELIMINATION product. Hint: Review…
A:
Q: What is the molar solubility of AgCl (Ksp = 1.80 × 1010) in 0.830 M NH₂? (Kf of Ag(NH3)2 is 1 x 107)
A: So, the molar solubility of AgCl in 0.830 M NH3 solution is approximately 3.44×106M.Explanation:…
Q: Which of the following mathematical expressions is the Henderson-Hasselbalch equation? 'a.¤ 'b.¤…
A: Answer Explanation:Step 1: Step 2: Step 3: Step 4:
Q: 3. Draw structural formulas for the a,ß-unsaturated aldehyde or ketone and the lithium…
A:
Q: Draw the major organic product formed in this reaction. HBr ROOR
A: Alkenes undergo free radical addition when treated with HBr in the presence of peroxide.This…
Q: The following H-NMR spectrum (showing all available signals) is for one of the possible products…
A: A triplet at 1.1 ppm and a quartet at 2.4 ppm suggests that the molecule has -CH2CH3 group. The…
Q: Ν 'N' SO₂R Bu3SnH, AIBN N SO₂R
A: The given reaction proceeds through the radical reactive mechanism. AIBN and Bu3SnH are used for the…
Q: Identify the geometry of each alkene as E, Z, or neither (non-stereoisomeric). HO X and Olis Z; Il…
A:
Q: 11. Which of the following molecules is a meso compound? CI All are meso None are meso A B C D E
A: Organic compounds can be defined as compounds that contain carbon and hydrogen atoms. We have been…
Q: 10.51 Draw all stereoisomers formed in each reaction. a. Br₂ b. Cl₂ ن H₂O NBS d. DMSO, H₂O H₂O H2SO4
A: As images cannot be uploaded to this section, see below for the answers:Explanation:The answers are…
Q: What is the equilibrium formula for the formation of bicarbonate from carbon dioxide and water?…
A: The formation of bicarbonate from carbon dioxide and water -We have to write the equilibrium formula…
Q: 1 sentence justification of your answer for each item. Without doing any calculations, determine the…
A: Entropy (S) is the degree of randomness.A positive sign of S indicates an increase in entropy and a…
Q: Consider the reaction: CO2(g) + CCl4(g) ⇌ 2 COCl2(g) Calculate ΔG for this reaction at 25 °C under…
A: CO2 (g) + CCl4 (g) → 2COCl2 (g)∆G° = 2∆G° (COCl2) - [ ∆G°f (CO2) + ∆G°f (CCl4)] =…
Q: And Option E is tsoh, h2o S ง 2 CHISH,H B HOCHUCHUCHUGH
A: When an aldehyde reacts with propane-1,3-dithiol in presence of acid catalyst it forms the…
Q: What is the photon energy of light with a wavelength of 399 nm? Note that h = 6.626 × 10-34 J's and…
A: Given:Wavelength of light = 399 nm = 399 x 10-9 m = 3.99 x 10-7 mPlanck's constant (h) = 6.626 x…
Q: 2.93 g Measured Volume of Water: 100.0 mL each question with correct significant figures and units.…
A: Given,Measured mass of NaCl = 2.93 gMeasured Volume of Water = 100.0 mLmolar mass of NaCI = 58.44…
Q: An analytical chemist is titrating 109.4 mL of a 0.7700 M solution of hydrazoic acid (HN3) with a…
A: We will first calculate the millimoles of both acid and base. Then we will find whether weak acid or…
Q: Which option contains the most acidic proton? ६ ६ ४ ६ a. b. C. d.
A: >A-H ---> A- + H+Greater the stability of conjugated base (A-) stronger is the conjugate…
Q: [B] (M) A reaction between substances A and B has been found to give the following data: [A] (M)…
A: Rate law is an expression which relates rate of reaction, Rate constant and concentration of…
Q: Identify the following molecule. CH₂OH Он HO Он Он
A: Given organic compound structure.we need to identify the given organic compound structure.
Q: Which group when attached to the benzene ring does not oxidizes by hot permanganate in the same way…
A: Given question is based on oxidation reaction.The alkyl side chain of benzene ring undergoes…
Q: Use VSEPR to predict bond angles about each highlighted atom. (a) H3C =1 N CH I :OH (b) H3C N H CH…
A: Answer:Molecular formulaHybridizationBond angleAX2E0sp1800AX2E1sp2Less than 1200AX2E2sp2Less than…
Q: Identify
A: Three salts form a basic solution in water,KNO2Basic solutionNa3PO4Basic solutionNH4CNBasic…
Q: Draw the product, please ง ง L. CR,CH-CH-Mg Dr 2. H₂O 1. HO 2. PCC J. CH,MgBr 4. H,O LHCN KCN 2.…
A: We ahve to predict the products.
Q: 10.38 Give the IUPAC name for each compound. a. b. C. d. نه f. ОН OH
A:
Q: 2. IN HNO3 H2SO4 % o/p/m?
A: Information about the question
Q: Question 6. Predict the product of the following reaction and draw the mechanism. Includes all…
A: In electrophilic aromatic substitution, the hydrogen of the benzene is replaced by an electrophile.…
Q: Predict the major product for the following reaction, include Stereochemistry of appropriat a. NaBH4…
A: The objective of the question is to predict the major product for the given reaction sequence. The…
Q: a. Both the reactant and product show very similar IR spectra. For the reactant, indicate which IR…
A: The objective of the question is to interpret the IR spectrum of the acetanilide and 4-bromo…
Q: ct Question 4 0 / 9 pts Sulfuric acid reacts with sodium hydroxide according to this reaction: H₂SO4…
A: The objective of the question is to calculate the molarity of the sulfuric acid (H2SO4) solution.…
Q: Provide an acceptable name for the following compound. 0- CH-N-CH2CH3 Spell out the full name of the…
A: RULES for IUPAC nomenclature; In first step, we identify functional groups present. We make longest…
Q: The concentration of the stock glucosamine solution is 100 mM. A student took 0.5 ml of this stock…
A: “Since you have posted multiple questions, we will provide the solutiononly to the first question as…
Q: 4) Determine the [H+] in a 0.755 M HCIO solution. The Ka of HCIO is 2.9 x 10-8. 2.9x 10-8 x2/0.155…
A: 4) 5)
Q: A chemist dissolved an 14.5-g sample of KOH in 100.0 grams of water in a coffee cup calorimeter.…
A: Given:Mass of KOH dissolved =14.5 gMass of water in a coffee cup calorimeter = 100.0 gTemperature…
Q: Propose a concise and efficient synthesis of the target molecule. Br steps starting material C…
A:
Q: Draw the structures of the products, showing stereochemistry where relevant. 3. a)…
A: These are the examples of electrophilic addition reactions.
Step by step
Solved in 3 steps with 2 images

- The following reaction has a ΔSsystem < 0 O2(g) → 2O(g) T or F can you explain why this is false?select the most appropriate reagent(s) to effect the change. K2Cr2O7, H+ H2, Pd 1. Disiamylborane, 2. HO–, H2O, H2O2 NaOCl H2SO4, HgSO4A hydrocarbon of unknown structure has the formula C8H10. On catalytic hydrogenation over the Lindlar catalyst, 1 equivalent of Η2 is absorbed. On hydrogenation overa palladium catalyst, 3 equivalents of Η2 are absorbed. (a) How many degrees of unsaturation are present in the unknown structure? (b) How many triple bonds are Present? (c) How many double bonds are present? (d) How many rings are present? (e) Draw a structure that fits the data.
- Provide the mechansim using curved arrows of the reaction of p-t-butyl phenol treated with acetic anhydride in AlCl3. Include resonance stabilized intermediates and if more than one product is formed, label them as major, minor, etc. thank you for the helpFor problem 8.17, all of the reactions will be SN2. For each reaction, identify and evaluate each nucleophile (strong? weak? Strong or weak as a base?) Also, evaluate each solvent as polar protic or polar aprotic. I recommend drawing the structure of each solvent. (g) Sodium methanethiolate (NaSCH3) in ethanolAttached compounds undergoes E2 elimination with strongbase? For compounds that undergo elimination, draw the product. Forcompounds that do not undergo elimination, explain why they are unreactive.
- Complete the following reaction sequences by drawing the major products or the reagents necessary to make them. Be sure to include stereochemistry when appropriate. Please answer fast I give you upvote(a) What happens when CH3—O—CH<sub3 is heated with HI?(b) Explain mechanism for hydration of acid catalyzed ethene :CH2 = CH2 + HzO CH3—CH,—OHWhat is the main product from the reaction sequence below?
- Match the questions with the correct descriptions below: Sn2, E2 E1cb Sn1, E1 (anti) E2, E1cb Sn1, E1 2 steps poor leaving group 2 carbons removed from a carbonyl group carbanion intermediate 3°>allylic, benzylic>2°>1°>CH₃ carbocation intermediate Ist order reaction (r=k[RX]) H-X anti no intermediate 2nd order reaction (r=k[RX][Nu]) polar aprotic solventplease complete the reaction. Write the main product of the reaction. Please answer fast i give upvotePlease state if the substrate below undergoes carbocation rearrangement in an SN1 mechanism Thank you



