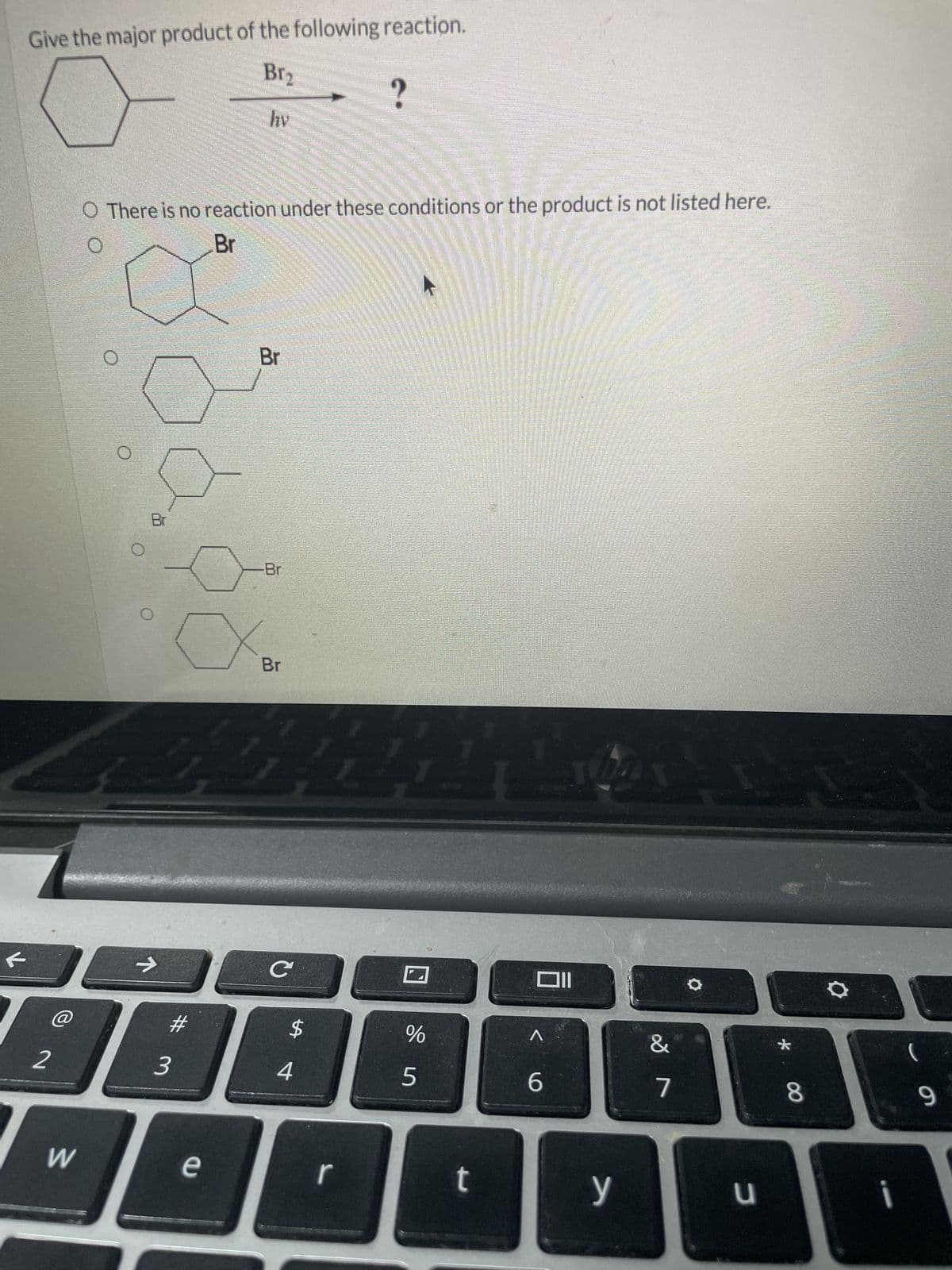
Give the major product of the following reaction. Br₂ hy O There is no reaction under these conditions or the product is not listed here. Br ← Br Br Br ✓ B Br -->> C וום @ # $ 2 3 4 55 % Λ & * 6 7 8 9 W e r t y כ
Q: Which of the following compounds is most acidic? A B C D
A: A-H ----> A- + H+The greater the stability of conjugate base (A-) the greater is the acidic…
Q: Give the name of the branched alkyl group attached to each of C4 alkyl groups, for example…
A: To name the branched alkyl group attached 1)Identify the longest chain of side chain2)Number the…
Q: Give Common name Write the common (not systematic) name of each organic molecule. structure CH3…
A: The given compound is a tertiary amine in which a nitrogen atom is attached with three alkyl groups.
Q: which of the following structures is aromatic (select all that apply)? N A B C D E F G H
A:
Q: 1) Rank the following acids in order of acidity (1 = most acidic, 4 = least acidic) CC13 ΘΗ ОН CF3…
A:
Q: Synthesis this please. Include reagents that could work and intermediate compound. N
A: The question is based on the concept of organic synthesis.We need to synthesize the product using…
Q: Draw one of the two enantiomers of the major product from this reaction. Use a dash or wedge bond to…
A: The addition of ICl to unsymmetrical alkene is takes place according to Markovnikovs rule. That…
Q: H₂C H NBS CH3 CCl4, hv H3C Br CH3 **You may assume that Br-Br is formed by a side reaction that…
A: The question is based on the concept of organic synthesis.We need to synthesize the product using…
Q: In the movie, the white and green bonds come together and pull apart, and they just move around the…
A: No chemical reaction taking place Explanation:If they are simply coming together and moving, there…
Q: Determine concentration of hydroxide ion, OH, in a solution of HNO₂ by constructing an ICE table,…
A: Answer:Upon adding in water weak acid gets partially ionized and an equilibrium between its ionized…
Q: answer the chemistry problem in the image and show solution step by step
A: The objective of the question is to complete the tables.
Q: Fill in the left side of this equilibrium constant equation for the reaction of hypochlorous acid…
A: Dissociation of HClO in water is given by:
Q: Is the following alkene s-cis ors-trans?
A: Geometrical isomerism is also called cis-trans isomerism, in which the different arrangements of…
Q: The molality of ethanol solution in which the mole fraction of water is 0.73 is:- (Hint:-Assume…
A:
Q: Write a balanced equation and the Kb expression for the Brønsted-Lowry base CH32NH in water. In the…
A: The balanced chemical equation represents the reaction of methylamine (CH2NH2) in water (H2O) to…
Q: 1. (a) Compound A,B and C are isomers with molecular formula of C4H8O. When compound A,B and C…
A: The objective of the question is to deduce the structures of compounds A, B, and C based on their…
Q: Provide the correct structure from the IUPAC name. (a) 2-aminocyclopropanecarboxylic acid (b)…
A: In the Bond line formula, a bond is represented by a single line.Only heteroatoms and hydrogen…
Q: HO NH₂ NH2
A: The objective of this question is to convert a given compound into a specified target compound…
Q: How does the NMR spectrum support the structure of Isoamyl Acetate
A: Nuclear Magnetic Resonance (NMR) spectroscopy is a powerful analytical technique used to determine…
Q: What size combinatorial library could be constructed using the substituents and diversity sites in…
A: The objective of this question is to identify substituents and diversity sites and calculate the…
Q: What is the optimum pH to separate a mixture of lysine, arginine, and cysteine using…
A: Electrophoresis is a technique that separates amino acids, relying on their net charge and the…
Q: Draw major E2 ELIMINATION product. Hint: Review Chapter 10.3. the elimination can only occur if the…
A:
Q: 12.20 Propose an efficient synthesis for each of the following transformations HO (a) JOH (b) Br…
A: The question is based on the concept of organic synthesis.We need to synthesize the product using…
Q: Calculate the pH at the equivalence point for the following titrations. 4A. Benzoic 50. mL of 0.12 M…
A: Given,4A) Molarity of benzoic acid = 0.12 Mvolume of benzoic acid = 50. mLMolarity of potassium…
Q: Select all of the α protons in the following two molecules that could be deprotonated by NaOH and…
A: Alpha proton
Q: Calculating the pH at equivalence of a titration 0/5 Izabella A chemist titrates 80.0 mL of a…
A: pH = 5.90Explanation:Given: V(CH3)2NH=80.0mL;[(CH3)2NH]=0.7536M;[HBr]=0.0940M;pKb=3.27at the…
Q: Draw the appropriate Newman projection which leads to the major E2 ELIMINATION product. Hint: Review…
A: E2 elimination reactions occur when the leaving group and hydrogen are anti-peri planar to each…
Q: Draw major E2 ELIMINATION product. Hint 1: Review Chapter 10.3, the elimination can only occur if…
A: Based on your question we need to find out the product by using beta elimination mechanismI'm…
Q: 6. Diprotic oxalic acid (HO2C−CO2H) ionizes in water as follows. HO2C−CO2H + H2O HO2C−CO2- + H3O+…
A: For part (b) 0.10 M solution of potassium oxalateOnly part B is required to be solved.
Q: The value of Kc for the reaction between water vapor and dichlorine monoxide is 0.0900 at 25°C.…
A:
Q: Calculate the mass of isoamy (isopentyl) alcohol corresponding to 0.045 mol and the volume of acetic…
A: The objective of the question is to calculate the mass of isopentyl alcohol.It is given that,The…
Q: At-9.93 °C the concentration equilibrium constant K = 6.2 for a certain reaction. Here are some…
A: The objective of the question is to answer the questions related to the information provided by the…
Q: How many π electrons are in the following compounds? A. B. C. D. IN:
A: We are given 4 molecules and we have to find number of π-electrons in each molecule.
Q: Draw the appropriate Newman projection which leads to the major E2 ELIMINATION product. Hint: Review…
A:
Q: Methanoic acid (HMeth) has a Ka of 1.78 * 10-6, so what is the pOH of a 0.23 M NaMethsolution?
A: The objective of this question is to calculate the pOH of a 0.23 M NaMeth solution, given that the…
Q: Which answer best describes the transfer of heat that occurs when 1.71 mol H, reacts with 0.814 mol…
A: Moles of H2 = 1.71 mol Moles of O2 = 0.814 mol Transfer of heat =?
Q: 4) Assume you have a solution 2.0M of HCIO that also contains 0.70M Bleach (NaCIO) Setup the…
A: The objective of the question is to determine the pH of a solution containing 2.0M of HCIO and 0.70M…
Q: A major HOH 人 ? minor B C D Η major 人 人 ΟΗ Хон major minor minor
A: SN1 and E1 Reaction : The SN1 and E1 reactions are unimolecular processes. This is a two-step…
Q: Provide the major expected products: MgBr 1. CO2(s) 2. HCI, H2O
A: The given starting compound is a Grignard regent( RMgX). Grignard Regent is aryl or alkyl magnesium…
Q: Predict the products of the following reaction. If no reaction will occur, use the NO REACTION…
A: A metal reacts with water to form metal hydroxide and hydrogen gas. Highly reactive metals can react…
Q: 12 10 8 Hd Hu 6 4 2 0 0 4 8 12 16 20 24 28 32 36 Volume Base Added (mL) 8A. What was in the…
A: A titrant is the solution which is added from burette while a sample is taken in titration flask.In…
Q: Choose the structure or structures that BEST represent the product for the following Diels-Alder…
A: The Diels–Alder reaction is a pericyclic reaction (cycloaddition) between a conjugated diene and a…
Q: 3. Consider a 2 × 2 square lattice of spins interacting via the Ising Hamiltonian in the absence of…
A: The objective of the question is to find all possible configurations of a 2x2 square lattice of…
Q: Each row of the table below describes an aqueous solution at about 25 °C. Complete the table. That…
A: Q. Given incomplete tableSolution[H3O+]pHAB9.34CHere in this question we have to complete the table…
Q: The preparations of two aqueous solutions are described in the table below. For each solution, write…
A: of is added to of a solution of is added to of a solution that is in both and We have to…
Q: Draw the major product of this reaction. Ignore inorganic byproducts. 1. CH3CH2CH2NH2, Ni, H2, pH…
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: Analyze and compare the spectra of the reactants and products in this Esterification chemical…
A: 1H-NMR spectroscopy is a spectroscopic method for the structural elucidation of a molecule. In a…
Q: Examine the model reaction below. CH,OH + → Cl + HCI OCH3 Assuming that the reactants in the second…
A: Final answer is given in explanation please see from there.Explanation:Approach to solving the…
Q: 20 20. Draw two reasonable resonance structures for the structure shown below H
A: The objective of the question is to find the two resonance structures of the given…
Q: What is the major product of the following reaction? III H
A: This is an example of enamine formation reaction
Step by step
Solved in 1 steps with 2 images


