Q: Given: 0.35g NaCl, 0.25 g NaHCO3, 0.15 g KCl & 2 g C6H12O6 are present in 100 mL ORS solution (MW:…
A: Given: 0.35g NaCl, 0.25 g NaHCO3, 0.15 g KCl & 2 g C6H12O6 are present in 100 mL ORS solution…
Q: Give ionic and net ionic equations for the following (see picture)....
A: The actual balanced equation is given below.
Q: The bisulfide anion (HS') has a pk, of 19.0. This means that a 0.10 M aqueous solution of HS is…
A:
Q: What is the solubility of PbF2 in water? (Ksp of PbF2 is 3.6 ×
A: PF2 dissociate in water as : PF2 (aq)→ P+2 (aq) + 2F-1 (aq) The Ksp (solubility product)…
Q: what is the oxidation state of Al2Se3
A: The compound given is Al2Se3.
Q: idae which are native to Colombia. identify the nybridiza он HC 10 6. 7. 8. 9. 10.
A: Hybridization is overlapping of atomic orbitals. In Hybridization Lone pairs and sigma bonds are…
Q: a. Cr (9) + H2O (0 the product will contain chromium(III) b. AgNO3 (aq)+ LİOH (aq)
A: 4. a) • Chromium metal reacts with water to produce Chromium(III) oxide and hydrogen gas.…
Q: At a particular temperature, 4.08 g of Gd2(SO4)3 will dissolve in 250 mL of water. What is the Ksp…
A:
Q: What are the products of the following reaction? O OH OH concentrated HI and and and and HO K HO
A: -> In presence of HI ether can break and form alcohol and alkyl iodide .
Q: 19.Which explanation below best describes why the following reaction occurs? 3Zn + H33PO44 --> 3H22…
A:
Q: Thallium is a very toxic element. Ion Tl+ is prone to form strong compounds with sulfur- containing…
A:
Q: A 0.9056 g sample of KBrO3 (MM=167.0) was dissolved in dilute HCl and treated with an unmeasured…
A: Moles of KBrO3 = 0.9056/167 = 0.00542 Moles of thiosulphate required = 6*0.005432 = 0.0325
Q: How does the reaction of ROH with HX occur?
A: In the reaction between an alcohol i.e ROH and HX, the below steps takes place. (The reaction…
Q: What are the respective concentrations (M) of Fe3+ and I- afforded by dissolving 0.200 mol FeI3 in…
A: Molarity is defined as moles of solute per liter volume of solution. It is represented as M. M=no.…
Q: ntify the type of reaction for the che ation shown: + 2BCI3 3CaCl2 + B
A: The question is based on concept of chemical reaction. we have to identify what kind of reaction is…
Q: Which of the following is the structure of oxalic acid? HO. HO. HO. HO. HO. II II IV B) I| OA)I O D)…
A: Answer The stracture of oxalic acid(C2H2O4) is
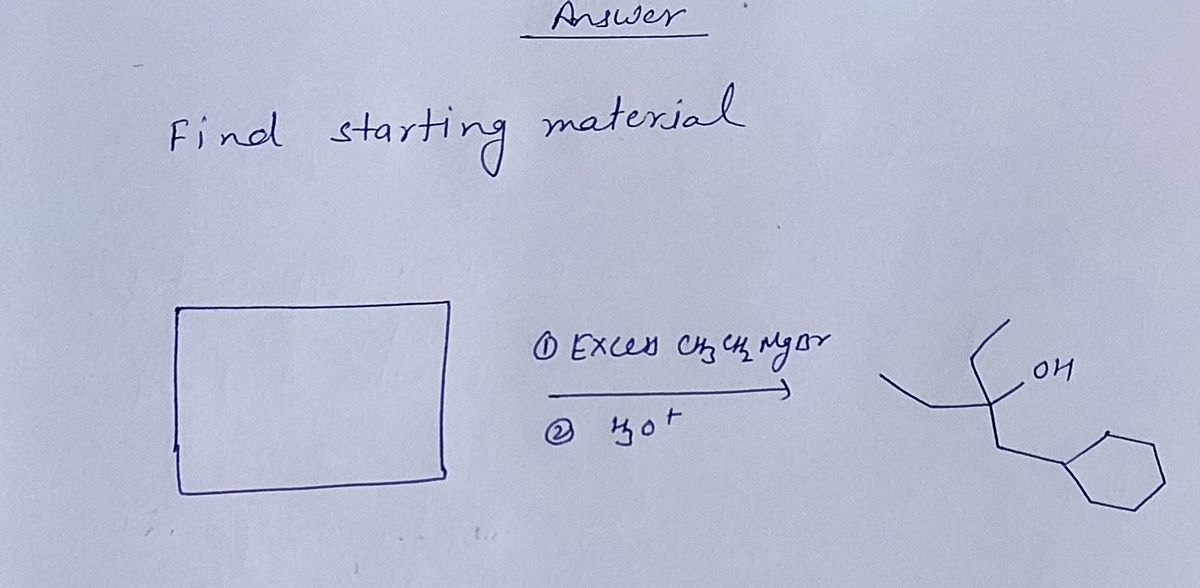
Q: (i) Me3Sn. OEt Pd(PPH3)4/CO A (ii) Br Ph Pd(PPH3)4, KOH -B(OH)2 В Br +
A:
Q: A 0. 4054 g solid organic sample containing covalently bound bromide and no other halogens was…
A: Given that : The mass of the solid organic sample = 0.4054 g The mass of AgBr produced = 37.8 mg The…
Q: When 55.5 g of calcium chloride CaC12 was dissolved in 0.5 L of water, so the concentration of the…
A: Given Mass of CaCl2 = 55.5 gram Volume = 0.5 Liter Molarity = ?
Q: Use data from CRC_Std_Thermodyn_Substances and CRC_Std_Thermodyn_Aqueous-Ions to calculate ΔrH∘ΔrH∘…
A: Given reaction is : 2CaC2O4 (s) ⟶ 4CO (g) + O2 (g) + 2CaO (s) , ΔrH∘ = ? Calculate the values of…
Q: Complete, balance and determine the type of the following reactions: A) C2H6S (l) + O2 (g) →…
A: In order to balance any chemical equation, we must ensure that the number of individual atoms on…
Q: What is the oxidation number of C in K2C2O4?
A:
Q: A sample of processed meat scrap weighing 4.000 g is digested with concentrated HSO. and…
A: The solution of the given question is
Q: coefficient for H2O(l)
A:
Q: 1. A mixture containing only KCl and NaBr is analyzed by the Mohr Method. A 0.3172-g sample is…
A: Given that : The mass of the sample = 0.3172 g The volume of water = 50 mL The volume of AgNO3 used…
Q: the aqueous solution of KNO2 has the following environment: a) acid b) neutral c) basic d) same as…
A: The aqueous solution of KNO2 is
Q: Among 5 different species (VO,*, Vo2*, v³*, v²*, v), how many are stable in water under pH=0? Acidic…
A: This diagram ( at pH= 0) just give you with ordinary value only for conceptual basis.Here we can see…
Q: Explain The Relationship between ?G°rxn and K?
A: ΔG° is defined as the change in standard Gibbs free energy. K is the equilibrium constant and is…
Q: How many grams of potassium citrate (C6H5K3O7 * H2O - mw 324) should be used in preparing 500 mL of…
A: Moles of solute per unit volume is known as Molarity
Q: 23. Which substance below exhibits the weakest IMFS? H2O CO2 H2CO SiO2
A: Q 23. London Dispersion forces exhibits weakest IMFs is:
Q: What is observed when the Na2SO3 solution is added to the CuCl2 solution?
A: Given question is : What is observed when the Na2SO3 solution is added to the CuCl2 solution = ?
Q: 4) Will the following reaction occur? Explain. (pKa of H20 is 14, pKa of C2H2 is 25, pKa of HCOOH is…
A: (a) No, this reaction will not occur. Reason: H2O is stronger acid than C2H2 so, C2H- ion is…
Q: electrolyte, ölytewhenitis dissolved in water? What species are present inC12H22O11(ag)?
A: The question is based on the concept of solutions. We have to identify nature of sucrose as an…
Q: IF 250 ml of a 15%(viv) solution of methyisalicylate in alcohol is diluted to 750ml, what will be…
A: Given Volume of solution ( V1 ) = 250 mL Initial %(V/V) = 15 % Final Volume of solution ( V2 ) =…
Q: Describe in words how you do the following preparation. Then give molecular equation for the…
A: The common sources of calcium carbonate are eggshells, marbles and chalk. Calcium acetate can be…
Q: Calculate the final molarity of H2O2H2O2 if 5.1 mL5.1 mL of a 3.0% w/w H2O23.0% w/w H2O2 solution,…
A: H203 solution is3.0 5 so 100 g of h202 solution contain 3 g calculate volume of 100 g solution…
Q: A 0.1017 g sample of KBrO3 (MM=166.1) was dissolved in dilute HCI and treated with an unmeasured…
A:
Q: Based solely on the amount of available carbon, how many grams of sodium oxalate, NazC2O4. could be…
A:
Q: A3B5 and a Ksp of 7.6E-26.
A: The pB value for given conditions is 3.75
Q: What is the oxidation number for the S in BaS2O3?
A:
Q: 20- Brown rinc In a salt Experim (a) Nitrates are (b) Fe2* acts as (C) Brown ring (d) NO gas forn a
A: Brown ring test: It is also known as the nitrate test. This test is performed to check out the…
Q: how many grams of BaF2 molar mass= 175.337 will dissolve in 300 mL of 0.30 M NaF solution? the ksp…
A:
Q: CN a. CN b. CO,Me C.
A:
Q: ng is not indicated by this reaction? KI, H;PO4 ether, 25° Č
A:
Q: In the following equation: HNO2+ NaHCO3 (yields) NaNO2+H2O+CO2 Why does this result in water…
A: Gas forming reaction: The reaction of acid and metal carbonates which produce carbonic acid. The…
Q: What is the oxidation number of X in H3XO3?
A:

Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- PHOTOSYNTHETIC PIGMENTS 1.6 0.7 Qy D 1.4 CH. 0.6 1.2 Soret 0.5 A N- 0.4 0.8 H COOCH3 0.3 0.6 phytyl bacteriochlorophyll a 0.2- 0.4 0.1 0.2 - 300 400 500 600 700 800 700 750 800 850 900 2 (nm) 2 (nm) Figure 4.7 Absorption (left) and fluorescence (right) spectra of bacteriochlorophyll a in diethyl ether. 5. Look at this figure from Blankenship's book, Molecular Mechanisms of Photosynthesis. You can see the structure for bacteriochlorophyll a, along with its absorption spectrum. a. What is the molecular shape around the labeled carbon atom? b. What is the hybridization around that same carbon? c. Based on this structure, would you expect this molecule to absorb light in the visible spectrum? Why or why not? Absorbance Fluorescence (Arbitrary units)1.) PBr3 2.) NaCN 3.) MgBr OH 4.) H*, H20 a) b.) d.) IZ ZI1. LIAIH4 a. 2. H;O* b. H2NNH2 КОН c. (CH;CH2)½CULI Cl d. (CH3)NH2 e. KMNO4, H3O* f. 1. DIBAH 2. H30*
- Which one of the following cannot be used as a deodorant? 2. a. CaCo3 b. Zn(C17H35CO0)2 C. Al(OH)3 d. Zno Which one of the following is not a suitable disinfectant against corona virus? a. Ethanol solution b. NaAl(OH)4 solution c. Phenol solution d. NaCIO solution Which one of the following phrases is not true? a. Acidic media do not affect soapless soaps b. Synthetic detergents are stronger than soaps C. Synthetic detergents cannot be used in hard water d. Alkylbentene sulphonates are synthetic detergents Dettol is: a. Antifertility. b. Antis ptic. C. Antipyretic. d. Antibiotic. Which one of the following is not an antacid? a. Mg(OH)2 b. CH3COONa c. Milk d. NaHCO3CO₂CH3 NH₂ CH₂(CH₂)2NH2 (excess) 1. P₂O5, A 2. CH3CH₂CH₂Li 3. H303 CF3 IZ H .N. & O 1. , H3O+ 2. NaOH, H₂O trifluoroperoxyacetic acid 3. 1. NH2NH2, NaOH, H2O, heat 2. KMnO4, NaOH, A 3. H3O*

