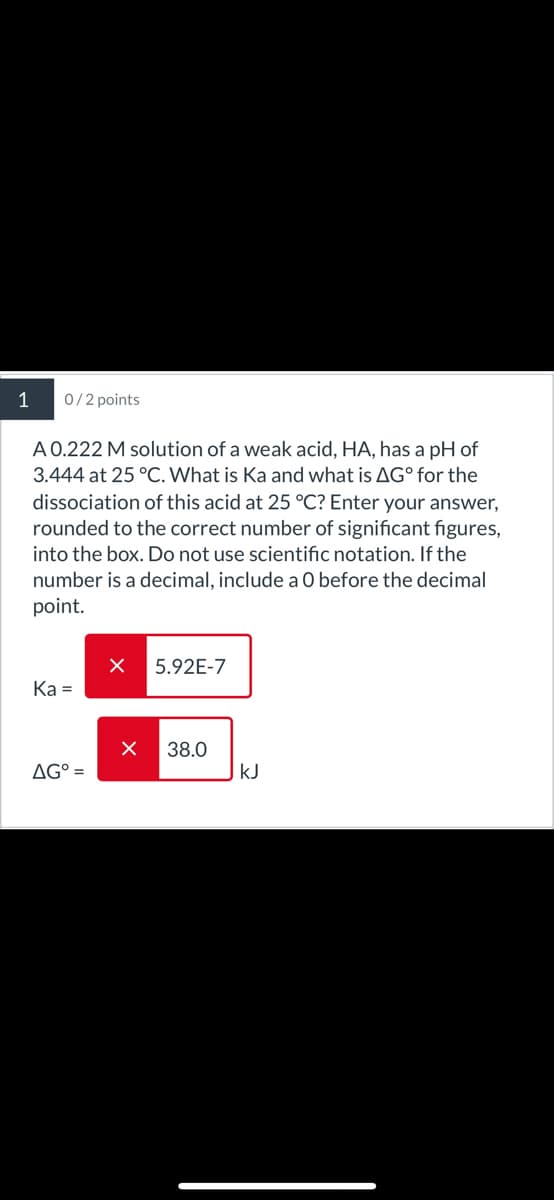
1 0/2 points A 0.222 M solution of a weak acid, HA, has a pH of 3.444 at 25 °C. What is Ka and what is AG° for the dissociation of this acid at 25 °C? Enter your answer, rounded to the correct number of significant figures, into the box. Do not use scientific notation. If the number is a decimal, include a O before the decimal point. 5.92E-7 Ka= ☑ 38.0 AG° = KJ
1 0/2 points A 0.222 M solution of a weak acid, HA, has a pH of 3.444 at 25 °C. What is Ka and what is AG° for the dissociation of this acid at 25 °C? Enter your answer, rounded to the correct number of significant figures, into the box. Do not use scientific notation. If the number is a decimal, include a O before the decimal point. 5.92E-7 Ka= ☑ 38.0 AG° = KJ
Chapter14: Acids And Bases
Section: Chapter Questions
Problem 162CWP: For solutions of the same concentration, as acid strength increases, indicate what happens to each...
Related questions
Question
Help me solve this.
Transcribed Image Text:1
0/2 points
A 0.222 M solution of a weak acid, HA, has a pH of
3.444 at 25 °C. What is Ka and what is AG° for the
dissociation of this acid at 25 °C? Enter your answer,
rounded to the correct number of significant figures,
into the box. Do not use scientific notation. If the
number is a decimal, include a O before the decimal
point.
5.92E-7
Ka=
☑ 38.0
AG° =
KJ
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning


Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning