
Which of the following would you expect to be more soluble in water than in benzene? (Select all that apply.)
(a)
(b)
(e)
Interpretation:
The compounds that is more soluble in water than benzene is/are to be identified.
Concept introduction:
The various kinds of interactions that bind a molecule are known as intermolecular forces. These can be dispersion, dipole–dipole, ion–dipole,and hydrogen bonding.
The solubility of a compound depends on the intermolecular forces present. Apolar solvent is soluble in apolar solvent only. A compound is soluble in water only when it is able to form hydrogen bonds with it.
Answer to Problem 1KSP
Solution: The correct answersareoption (a) and option (e).
Explanation of Solution
Reason for the correct option:
The molecule, methanol
Thus, it is soluble in water more readily. On the other hand, benzene is a nonpolar molecule. So, methanol is insoluble in benzene.
Potassium iodide
Hence, option (a) and option (e) are correct.
Reasons for the incorrect options:
Option (b) is incorrect because
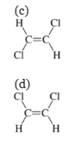
Option (c) is incorrect because trans-dichloroethene is a nonpolar molecule. It is insoluble in water. So, this option is incorrect.
Option (d) is incorrect because cis-dichloroethene is a polar molecule but it is unable to form hydrogen bonds in water. It is, therefore, insoluble in water. So, this option is incorrect.
Hence, options(c) and (d) are incorrect.
Want to see more full solutions like this?
Chapter 11 Solutions
Chemistry
- Consider a series of compounds: Methane (CH3), Ethane (CH3 CH3), Propane (CH3 CH2 CH3), Butane (CH3 CH2 CH2 CH3), and Pentane (CH3 CH2 CH2 CH2 CH3). For each compound (a) Indicate whether its molecules are polar or non polar. (b) Indicate predominant type of intermolecular forces in the liquid state. (c) Calculate the molar mass. (d) Convert each boiling point from Celsius degrees to Kelvins. (e) Plot a graph of boiling point (in K) versus molar mass. (f) Show the relationship between molar mass and boiling point?arrow_forwardBased on the type or types of intermolecular forces, predictthe substance in each pair that has the higher boiling point:(a) propane (C3H8) or n-butane (C4H10), (b) diethyl ether(CH3CH2OCH2CH3) or 1-butanol (CH3CH2CH2CH2OH),(c) sulfur dioxide (SO2) or sulfur trioxide (SO3), (d) phosgene(Cl2CO) or formaldehyde (H2CO).arrow_forwardWhich of the following gases is expected to be most soluble in water? Explain your reasoning.(a) CH4(b) CCl4(c) CHCl3arrow_forward
- True or false: (a) CBr4 is more volatile than CCl4. (b) CBr4has a higher boiling point than CCl4. (c) CBr4 has weakerintermolecular forces than CCl4. (d) CBr4 has a highervapor pressure at the same temperature than CCl4.arrow_forwardIf water were a linear molecule, (a) would it still be polar, and (b) would the water molecules still be able to form hydrogen bonds with one another?arrow_forwardList the substances Ar, Cl2, CH4, and CH3COOH in order ofincreasing strength of intermolecular attractions.(a) CH4 6 Ar 6 CH3COOH 6 Cl2(b) Cl2 6 CH3COOH 6 Ar 6 CH4(c) CH4 6 Ar 6 Cl2 6 CH3COOH(d) CH3COOH 6 Cl2 6 Ar 6 CH4(e) Ar 6 Cl2 6 CH4 6 CH3COOHarrow_forward
- Would Octane C8H18, be more soluble in ethanol C2H5OH or in benzene C6H6?arrow_forwardstrongest intermolecular force stated above. Explain how does high temperature affect the surface tension. (a) Diamond is an allotrope of carbon. Draw the lattice struarrow_forwardList the most important (strongest) intermolecular force (s) that must be overcome to (a) vaporize liquid SO 2 (b ) remove water of hydration from CaSO 4 • 2H 2 O (c) melt solid Al 2 O 3arrow_forward
- (a) 4. Identify the intermolecular forces which can operate between molecules of the following pure compounds: (b) N. (c) Numbering N. (d) 5. For the compounds in the previous question, what intermolecular forces would operate between the compound and water?arrow_forwardName the type(s) of intermolecular forces that exists between molecules (or basic units) in each of the following species and identify the following species that are capable of hydrogen-bonding among themselves. (a) BeH2, (b) CH3COOHarrow_forwardRank the following in order of decreasingsurface tension ata given temperature, and explain your ranking:(a) CH₃OH(b) CH₃CH₃(c) H₂C=Oarrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning

