
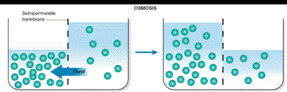
Assume that two liquids are separated by a semipermeable membrane, with pure solvent on the right side and a solution of a solute on the left side. Make a drawing that shows the situation after equilibrium is reached.
Interpretation:
The situation after equilibrium is reached when two liquids are separated by a semipermeable membrane has to be drawn.
Concept introduction:
Colligative properties such as vapor pressure, freezing point, and boiling point are affected by the presence of solute particles in a solution.
Osmosis is the passage of substances in true solution through a semipermeable membrane.
Particles such as starches and protein molecules are too large to pass the membrane.
The restriction of passage of large particles causes the smaller particles to pass more rapidly in the direction of higher concentrations, producing an osmotic pressure in confined liquids.
Explanation of Solution
Osmosis occurs when solution of different concentration are separated by a semipermeable membrane that allows solvent molecules to pass but blocks the passage of solute ions and molecules. Solvent flows from the more dilute side to the more concentrated side until sufficient osmotic pressure builds up and stops the flows. An effect similar to osmosis occurs when membranes of large pore size are used.
The situation after equilibrium is reached when two liquids are separated by a semipermeable membrane is drawn as
Want to see more full solutions like this?
Chapter 9 Solutions
Fundamentals of General, Organic, and Biological Chemistry (8th Edition)
- for 0.9% NaCl solution and 10% NaCl solution, which one has higher concentration of WATER?arrow_forwardAssume that you have a stock solution of 10% SDS and you need to make 150 microliters of 0.5% SDS. What volume of 10% SDS and what volume of water would you use?arrow_forwardDefine the term Tonicity of Solutions?arrow_forward
- Calculate the effective quantity (g) of sodium chloride related to tonicity in 100ml of an intravenous fluid labeled "5% dextrose in 0.45% sodium chloride," and indicate whether the solution is isotonic, hypotonic or hypertonic. The answer is Hypertonic and 1.35g NaCl but I don't understand how to get the answer.arrow_forwardWhen 2.43 g of a nonelectrolyte solute is dissolved in water to make 435 mL of solution at 25 °C, the solution exerts an osmotic pressure of 895 torr. What is the molar concentration of the solution? concentration: 0.045 M Incorrect How many moles of solute are in the solution? moles of solute: 0.034 mol Incorrect What is the molar mass of the solute? molar mass: 69.12 g/mol Incorrectarrow_forwardA 0.10 M NaCl solution is (hypertonic/hypotonic/isotonic) to a 0.1M glucose solution.arrow_forward
- Below is a Beer's Law Plot for the concentration of a specific colored compound x. A solution of compound X is measured with the spectrophotometer to have an optical density of 0.60. a) What is the concentration of compound X in this solution. Answers must contain proper units for full credit. b) The absorbance of this compound was measured at 540 nm. Why was this absorbance chosen? 0.9 0.8 0.7 0.6 805 4 0.4 0.3 0.2 Absorbance at 540 marrow_forwardGive detailed Solution with explanation (no need Handwritten answerarrow_forwardA mixture of lipids containing phosphatidic acid, cholesterol, testosterone, and phosphatidylcholine was applied to a hydrophobic interaction chromatography column. The column was washed with a high salt buffer and the lipids were eluted with decreasing salt concentrations. In what order would the lipids be eluted from the column? Explain your answer.arrow_forward
- Would a sample solution that has a high absorbance reading, have a low or a high concentrationarrow_forwardShow solution please.thank youarrow_forwardThe standard curve to determine the amount of betacyanin is shown below. You extracted a red pigment from a beet disc (the mass of a disc is 2 g) using 10 ml of 20% ethanol solution. You measured absorbance of the solution above the beet disc every minute for a total time of 20 minutes. The increase in absorbance was linear during a period of time from 1 min to 10 min. The absorbance at 10 min was 0.8. Calculate the amount of betacyanin extracted from 1g of a beet tissue per minute. Explain your calculations. You can use Excel or a calculator.arrow_forward
