
Concept explainers
Draw the product formed when the following amino acid is treated with each reagent:
(a)
(a)
Interpretation: The products formed by the treatment of given amino acid with
Concept introduction: The chemical compounds in which carbon is bonded with acidic and basic group along with hydrocarbon side chain are known as amino acids. The amine and carboxyl group of amino acid shows different reactions with alcohols, acid chloride, acid and base.
Answer to Problem 27P
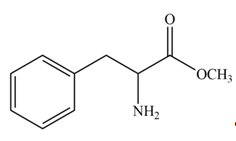
The products formed by the treatment of given amino acid with
Explanation of Solution
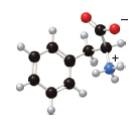
In the given ball-and-stick model of amino acid, the black balls represent carbon atoms, the white balls represent hydrogen atoms, the blue ball represents nitrogen atom and the red balls represent oxygen atoms. Therefore, the structure of given amino acid is,

Figure 1
The protection of carboxyl group takes place on reaction with
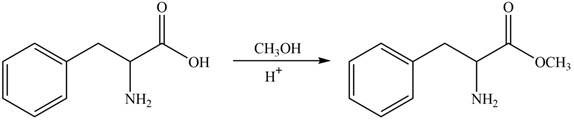
Figure 2
The products formed by the treatment of given amino acid with
(b)
Interpretation: The products formed by the treatment of given amino acid with
Concept introduction: The chemical compounds in which carbon is bonded with acidic and basic group along with hydrocarbon side chain are known as amino acids. The amine and carboxyl group of amino acid shows different reactions with alcohols, acid chloride, acid and base.
Answer to Problem 27P
The products formed by the treatment of given amino acid with
Explanation of Solution
The structure of given amino acid is shown in Figure 1.
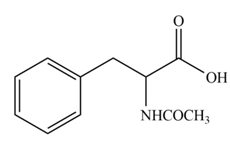
The amine group of amino acid form amide bond on reaction with acid chloride as shown in Figure 3.
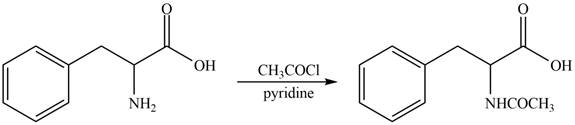
Figure 3
The products formed by the treatment of given amino acid with
(c)
Interpretation: The products formed by the treatment of given amino acid with
Concept introduction: The chemical compounds in which carbon is bonded with acidic and basic group along with hydrocarbon side chain are known as amino acids. The amine and carboxyl group of amino acid shows different reactions with alcohols, acid chloride, acid and base.
Answer to Problem 27P
The products formed by the treatment of given amino acid with
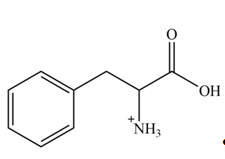
Explanation of Solution
The structure of given amino acid is shown in Figure 1.
The amine group of amino acid form amide bond on reaction with acid chloride as shown in Figure 4.
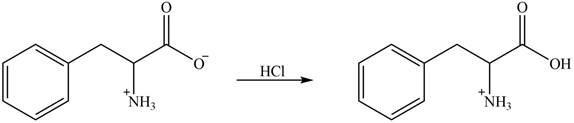
Figure 4
The products formed by the treatment of given amino acid with
(d)
Interpretation: The products formed by the treatment of given amino acid with
Concept introduction: The chemical compounds in which carbon is bonded with acidic and basic group along with hydrocarbon side chain are known as amino acids. The amine and carboxyl group of amino acid shows different reactions with alcohols, acid chloride, acid and base.
Answer to Problem 27P
The products formed by the treatment of given amino acid with
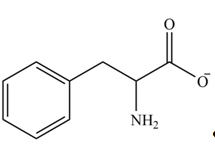
Explanation of Solution
The structure of given amino acid is shown in Figure 1.
The amine group of amino acid form amide bond on reaction with acid chloride as shown in Figure 5.
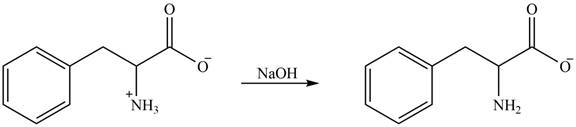
Figure 5
The products formed by the treatment of given amino acid with
(e)
Interpretation: The products formed by the treatment of given amino acid with
Concept introduction: The chemical compounds in which carbon is bonded with acidic and basic group along with hydrocarbon side chain are known as amino acids. The amine and carboxyl group of amino acid shows different reactions with alcohols, acid chloride, acid and base.
Answer to Problem 27P
The products formed by the treatment of given amino acid with
Explanation of Solution
The structure of given amino acid is shown in Figure 1.
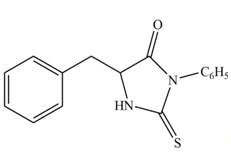
The terminal amine group of amino acid on reaction with phenyl isothiocyanate form N-phenylthiohydantoin as shown in Figure 6.
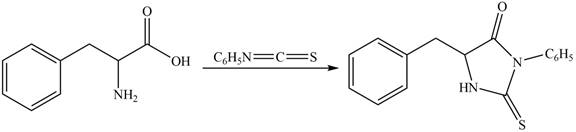
Figure 6
The products formed by the treatment of given amino acid with
Want to see more full solutions like this?
Chapter 27 Solutions
Organic Chemistry (6th Edition)
- 2. Classify the following amino acids as nonpolar, polar basic, polar acidic, or polar neutral. (a) (b) (c) (d) H₂N-CH-COH CH-OH I CH3 || H₂N-CH-C-OH I CH₂ C=O 1 OH H₂N-CH-C-OH CH₂ T CH-CH3 CH₂ H₂N-CH-C-OH ī CH₂ OH 229arrow_forwardwrite the IUPAC names for the following aminos CH,CH, CH;CH,NH2 b) CH;CH, c) CH 9. Escriha la aetunt -.arrow_forwardDraw the structures of the amino acids formed at physiological pH when the tripeptide below is hydrolyzed. Н Н 0 Н |+ 1 || | H-N-с-с-N-с Н H C CH Н ОН н с Н Н || | | C-N-C-C-0 CH, HC CH Нc CH c=0 с NH₂ ОНarrow_forward
- (a) What is the difference between the hormones progesterone and testosterone? (b) Draw the structure of a a steroid nucleus. (c) Give the products obtained from complete base hydrolysis in the following reaction: O || CH,−O−C−(CH2)14–CH3 O CH–0–C−(CH2)14—CH3 + 3 NaOH O CH,−0–C−(CH2)14–CH3arrow_forwardWhich of the following amino acids is naturally occurring? - CH,OH H- ČH,CO0- -NH, ČH,CH,COo-arrow_forwardDraw the amino acids and peptide fragments formed when the decapeptide A–P–F–L–K–W–S–G–R–G is treated with each reagent or enzyme: (a) chymotrypsin; 8 pt helvetica roman (b) trypsin; (c) carboxypeptidase; (d) C6H5N = C = S.arrow_forward
- / 3. Show how the following two amino acids form a protein (a dipeptide). CH3 CH,SH H,N-C-C-OH → || H,N–C-C-OH но ноarrow_forwardWhich of the follawing amino auids would be Classi hied as a Polar Neutral amino? 1. H A3NT.C-COOH 2. H H3N+-C-C00- CH2 CO0- 3. H HgNt-C-Co0- CH2 OH 4. H HgN+-C-CO0- CH3arrow_forwardDraw the structures of the amino acids formed at physiological pH when the tripeptide below is hydrolyzed. но HC ннон нонн с H-N-C-C-N-C-C-N-с-с H C. HC. || CH CH₂ =0 H₂N H C. SCH, o=c NH₂arrow_forward
- 5. Draw the all of the products formed when the following amino acids form a peptide bond through an Amidification reaction. NH, CH2 CH2 CH2 ČH2 CH2 H3N-C-COOH H2N-C-CO0 H. Harrow_forwardTreating chitin with H2O, -OH hydrolyzes its amide linkages, forming a compound called chitosan. What is the structure of chitosan? Chitosan has been used in shampoos, bers for sutures, and wound dressings.arrow_forwardThe following compound is a(n) CH200(CH2)14CH3 CHOC CH2)14CH3 monosaccharide triglyceride O harmone steriod dipeptide CH,OH CHOH CHOH is a OH H OH H OH H %3D H OH OH O polyamide O polypeptide O polyester O polysaccharide O polyurethanearrow_forward
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
