
Fundamentals of General, Organic, and Biological Chemistry (8th Edition)
8th Edition
ISBN: 9780134015187
Author: John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. Peterson
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 25, Problem 25.11UKC
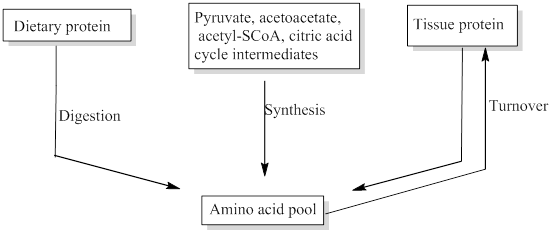
In the diagram shown here, fill in the sources for the amino acid pool.
Expert Solution & Answer
Interpretation Introduction
Interpretation:
The missing sources for the amino acids pool has to be filled.
Concept introduction:
- Amino acid had both amino functional group and carboxyl functional group in a molecule.
- Amino acid pool is the entire collection of free amino acids in the whole body.
Explanation of Solution
Free amino acids are present throughout the body, in cells and the extracellular fluids. This pool is supplied by few sources include,
- Non-protein nitrogen compounds
- Dietary protein
- Tissue protein etc.
Conclusion
The missing sources for the amino acids pool was filled.
Want to see more full solutions like this?
Subscribe now to access step-by-step solutions to millions of textbook problems written by subject matter experts!
Students have asked these similar questions
The image below shows an amino acid at physiological pH. Answer the following questions about
this amino acid.
H
|
//
- c
H3N*
|
CH2
CH2
|
H2N
a. How many carbons are in this amino acid's side chain? Entry Format: please type your answer as a
number. 3
b. Is the side chain of this amino acid ionized at physiological pH? Entry Format: Please type the
word yes or no without any capitalization. yes
c. At physiological pH, amino acids have two ionic charges, one positively charged group and one
negatively charged group. What is the term for this sort of molecule? Entry Format: Please type your
response with all lowercase letters.
Draw a diagram of a Ramachandran plot. Indicate the location on the plot of an
amino acid residue with phi = - 90 degrees and psi = + 120 degrees.
Are all the amino-acids soluble in water? Explain why?
Chapter 25 Solutions
Fundamentals of General, Organic, and Biological Chemistry (8th Edition)
Ch. 25.2 - Prob. 25.1PCh. 25.2 - Prob. 25.2KCPCh. 25.3 - Prob. 25.3PCh. 25.3 - Prob. 25.4PCh. 25.3 - Prob. 25.5PCh. 25.3 - Prob. 25.6PCh. 25.4 - Prob. 25.1CIAPCh. 25.4 - Prob. 25.2CIAPCh. 25.4 - Prob. 25.3CIAPCh. 25.4 - Prob. 25.7P
Ch. 25.4 - Prob. 25.8KCPCh. 25.6 - Prob. 25.9PCh. 25.6 - Prob. 25.10KCPCh. 25.6 - What is meant by a conditional amino acid?Ch. 25.6 - Prob. 25.5CIAPCh. 25.6 - Prob. 25.6CIAPCh. 25 - In the diagram shown here, fill in the sources for...Ch. 25 - Prob. 25.12UKCCh. 25 - Prob. 25.13UKCCh. 25 - Prob. 25.14UKCCh. 25 - Prob. 25.15UKCCh. 25 - Prob. 25.16UKCCh. 25 - Prob. 25.17APCh. 25 - Prob. 25.18APCh. 25 - Prob. 25.19APCh. 25 - Prob. 25.20APCh. 25 - Prob. 25.21APCh. 25 - Prob. 25.22APCh. 25 - What is the structure of the -keto acid formed...Ch. 25 - Prob. 25.24APCh. 25 - In general, how does oxidative deamination differ...Ch. 25 - Prob. 25.26APCh. 25 - Prob. 25.27APCh. 25 - Prob. 25.28APCh. 25 - Prob. 25.29APCh. 25 - Prob. 25.30APCh. 25 - Prob. 25.31APCh. 25 - Prob. 25.32APCh. 25 - Prob. 25.33APCh. 25 - Prob. 25.34APCh. 25 - How do essential and nonessential amino acids...Ch. 25 - Prob. 25.36APCh. 25 - Prob. 25.37APCh. 25 - How is tyrosine biosynthesized in the body? What...Ch. 25 - Prob. 25.39APCh. 25 - Prob. 25.40APCh. 25 - Prob. 25.41APCh. 25 - What energy source is used in the formation of...Ch. 25 - Write the equation for the transamination reaction...Ch. 25 - Prob. 25.44CPCh. 25 - Prob. 25.45CPCh. 25 - Prob. 25.46CPCh. 25 - Prob. 25.47CPCh. 25 - Prob. 25.48CPCh. 25 - Prob. 25.49CPCh. 25 - Prob. 25.50CPCh. 25 - Prob. 25.51CPCh. 25 - Prob. 25.52CPCh. 25 - Why might it be a bad idea to take large...Ch. 25 - Prob. 25.54GPCh. 25 - Prob. 25.55GPCh. 25 - Prob. 25.56GP
Additional Science Textbook Solutions
Find more solutions based on key concepts
1. Suppose a chloride ion and a sodium ion are separated by a center—center distance of 5 Å. Is
the interactio...
Biochemistry: Concepts and Connections
1. Suppose a chloride ion and a sodium ion are separated by a center—center distance of 5 Å. Is
the interactio...
Biochemistry: Concepts and Connections (2nd Edition)
Practice Problem ATTEMPT
Write the rate expressions for each of the following reactions:
(a)
(b)
(c)
Chemistry
7. (II) (a) What is the current in the element of an electric clothes dryer with a resistance of 8.6 ?when it i...
Physics: Principles with Applications
For each of the following 2-dimensional shapes, determine the highest order rotation axis of symmetry.
Inorganic Chemistry
Problem Set
True or False? Indicate whether each of the following statements about membrane transport is true (...
Becker's World of the Cell (9th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biochemistry and related others by exploring similar questions and additional content below.Similar questions
- Determine the pKa of the amino acid using the graph graph attached:arrow_forwardIdentify the amino acid that will give the following data:arrow_forwardSketch a titration curve for the amino acid cysteine, and indicate the pKavalues for all titratable groups. Also, indicate the pH at which this amino acid has no net charge.arrow_forward
- Which amino acids are in the figure?arrow_forwardGiven the amino acids below, what amino acid will elute first in a cation-exchange column. 1. Aspartate or Lysine using a buffer at pH 7 2. Arginine or Methionine using a buffer at pH 7 3. Lysine or Valine using a buffer at pH 7 4. Glycine or Valine using a buffer at pH 7 5. Serine or Alanine using a buffer at pH 7arrow_forwardIdentify the parts of the heterotrimeric G-protein shown in the image. Barrow_forward
- Consider the R groups of amino acids glycine, asparagine, and tyrosine. Note: Reference the Naturally occurring amino acids data table for additional information.arrow_forwardGive two examples of amino acids that are: Acidic Basic Polar Nonpolararrow_forwardGiven the Ramachandran Plot below, identify the protein components that could adopt the phi-psi angle combination indicated by the number 3. (IF YOU COPY UR ANSWER FROM ANOTHER WEBSITE I WILL DISLIKE)arrow_forward
- Provide the 20 amino acids in this format:arrow_forwardDraw a graph using optical densities of the amino acid ninhydrin complexes at different wavelengthsarrow_forwardUsing the data in the table below, calculate the average molar mass of an amino acid residue in each of the proteins in the table (excluding cytochrome c, myoglobin, and hemoglobin). Next, calculate the average molar mass considering all proteins (excluding cytochrome c, myoglobin and hemoglobin).arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

The Cell Membrane; Author: The Organic Chemistry Tutor;https://www.youtube.com/watch?v=AsffT7XIXbA;License: Standard youtube license