
Organic Chemistry (9th Edition)
9th Edition
ISBN: 9780321971371
Author: Leroy G. Wade, Jan W. Simek
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 21.2F, Problem 21.1P
Name the following carboxylic acid derivatives, giving both a common name and an IUPAC name where possible.
- a. PhCOOCH2CH(CH3)2
- b. PhOCHO
- c. PhCH(CH3)COOCH3
- d. PhNHCOCH2CH(CH3)2
- e. CH3CONHCH2Ph
- f. CH3CH(OH)CH2CN
- g. (CH3)2CHCH2COBr
- h. Cl2CHCOCl
- i. (CH3)2CHCOOCHO
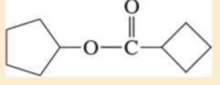
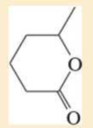
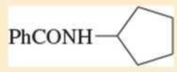
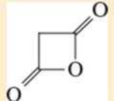
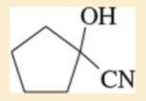
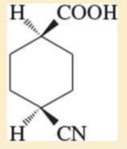
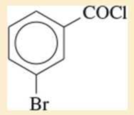
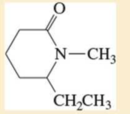
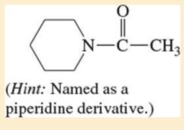
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Ch. 16 - Car
xylic AcluS
1.
Name the following carboxylic acids. Use both IUPAC and common names.
b) CHs-
cy-E-on
c) CHz-CHz-cooH
a)
--OH
d) CH-CHt
) CHy-CH と-OH
GOH
CH-CH-CHy
Ho-ビ-CH -
と。
f)
CH-CH-C-Cty
CH2
for e
Longth
k) Ho-
-CH
l CH
lee
Ho-C-C4,-CH-C-oH
Instructions: Draw out each compound to clearly show what groups are bonded to the carbonyl carbon.
Label each compound as a carboxylic acid, ester, or amide.
a. CH3CH2CO2CH2CH3
b. CH3CONHCH3
c. (CH3)3CCO2H
d. (CH3)2CHCON(CH3)2
Instructions: Give the IUPAC name for each compound.
A.
CH₂
CH₂CH₂CH₂CCH₂COOH
CH3
B.
CH₂CHCH₂CH₂COOH
CH₂COOH
CH₂CH3
C. (CH,CH,),CHCH,CHCOOH
Instructions: Give the structure corresponding to each IUPAC name.
a. 2-bromobutanoic acid
b. 2,3-dimethylpentanoic acid
c. 2-ethyl-5,5-dimethyloctanoic acid
d. 3,4,5,6-tetraethyldecanoic acid
• Whał arc the IUPAC namar of the ff. Carboxylic acias?
a.
COOH
COOH
CH3
b.
I
f.
.COOH
CHJCH2 ÇHCHCOOH
CH3
NO2
tON
C.
COOH
ноос
d.
COOH
Chapter 21 Solutions
Organic Chemistry (9th Edition)
Ch. 21.2F - Name the following carboxylic acid derivatives,...Ch. 21.4A - Prob. 21.2PCh. 21.4A - Prob. 21.3PCh. 21.4A - Prob. 21.4PCh. 21.5C - Prob. 21.7PCh. 21.6 - When ethyl 4-hydroxybutyrate is heated in the...Ch. 21.6 - Propose a mechanism for the following ring-opening...Ch. 21.6 - Prob. 21.15PCh. 21.7B - Prob. 21.16PCh. 21.7C - Prob. 21.19P
Ch. 21.7C - Prob. 21.20PCh. 21.7C - Prob. 21.21PCh. 21.7D - Prob. 21.22PCh. 21.7D - The mechanism for acidic hydrolysis of a nitrile...Ch. 21.8A - Prob. 21.24PCh. 21.8C - Prob. 21.25PCh. 21.9 - Prob. 21.26PCh. 21.9 - Prob. 21.27PCh. 21.9 - Prob. 21.28PCh. 21.10 - Draw a mechanism for the acylation of anisole by...Ch. 21.10 - Prob. 21.30PCh. 21.11 - Prob. 21.31PCh. 21.11 - Prob. 21.32PCh. 21.12 - Problem 21-33 Propose a mechanism for the...Ch. 21.12 - Suggest the most appropriate reagent for each...Ch. 21.12 - Show how you would synthesize each compound,...Ch. 21.13 - Prob. 21.36PCh. 21.13 - Prob. 21.37PCh. 21.14 - Prob. 21.38PCh. 21.14 - Prob. 21.39PCh. 21.16 - Prob. 21.40PCh. 21.16 - Prob. 21.41PCh. 21 - Prob. 21.42SPCh. 21 - Give appropriate names for the following...Ch. 21 - Predict the major products formed when benzoyl...Ch. 21 - Predict the products of the following reactions....Ch. 21 - Prob. 21.46SPCh. 21 - Prob. 21.47SPCh. 21 - Prob. 21.48SPCh. 21 - Propose mechanisms for the following reactions.Ch. 21 - Prob. 21.51SPCh. 21 - An ether extraction of nutmeg gives large...Ch. 21 - Prob. 21.53SPCh. 21 - Show how you would accomplish the following...Ch. 21 - Prob. 21.55SPCh. 21 - Prob. 21.56SPCh. 21 - Prob. 21.57SPCh. 21 - Prob. 21.58SPCh. 21 - Prob. 21.59SPCh. 21 - Explain this curious result. What does this...Ch. 21 - Prob. 21.61SPCh. 21 - Prob. 21.62SPCh. 21 - Prob. 21.63SPCh. 21 - A chemist was called to an abandoned aspirin...Ch. 21 - Prob. 21.67SPCh. 21 - The IR spectrum, 13ONTVTR spectrum, and 1HNMR...Ch. 21 - Prob. 21.69SPCh. 21 - Prob. 21.70SPCh. 21 - Prob. 21.71SP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Which is propyl propanoate? A. CH₂CH₂CH₂OOCCH₂CH; B. CH₂CH₂CH₂COOCH₂CH₂ C. CH₂CH₂CH₂COCH₂CH₂ D. CHỊCH,CH,OCH,CHỊCH, A B C Darrow_forwardWhat is the systematic IUPAC name for the given compound? CH3 CH3 CH;CHCH,CH,CH2 -Ń-CH3 a. N-methyl-4-methylhexan-1-amine b. 2,4-dimethylhexan-1-amine c. 2,2,N-trimethylpentan-1-amine d. N,N,4-trimethylpentan-1-aminearrow_forward614 Give the IUPAC name for each of the following 5-14 carboxylic acids. a. CH3-CH2-CH2-CH2-C-OH CH3 CH3 bilk b. CH—CH-СН,—С—ОН CH3 CH3 O c. CH3-CH-CH-C-OH CH-CH—С-ОН d. CH3-CH,—СH,—СООНarrow_forward
- Las o thist s Exercises and Próblems 5 16-20 Give the IUPAC name for each of the following carboxylic acids. a. HO HO, b. O CI HO-C-CH-CH2-CH2-C-OH C. СООН CH2-CH3 d. СООН CI 16-21 Draw a condensed structural formula that correspo to each of the following carboxylic acids. a. 2,2-Dimethylbutanoic acid h 2.2-Dimethylbutanedioic acidarrow_forwardWhat is the IUPAC name of the following compound? Br CH3 H3C O A. 5-Bromo-2-methylphenyl ethanoate B. 3-Bromo-6-methylphenyl ethanoate C. 4-Bromo-2-{oxy-(1-oxoethyl)} toluene EO D. Methyl-5-bromo-2-methyl benzoatearrow_forwardErythronolide B is the biological precursor of erythromycin, a broad-spectrum antibiotic. What functional group Erythronolide B does contain? a. b. H₂CH₂C C. H₂C 1 H₂C 2 3 4 O Amide d. Amine OH Erythronolide B Ketone Aldehyde a CH₂ b C d CH₂ OH JCH₂ 'OH OHarrow_forward
- H3C. H3C CH3 Br 1-propanolarrow_forwardPhenol is Select one: a. CH3CH2CH2OH b. C8H11OH c. CH3CH2CH2CH2CH2OH d. C6H5OHarrow_forwardI. Complete the table. Classification of Carboxylic Acids and its derivatives No. Common Name Structure IUPAC Name 1. CH3CH2CH2COONA COOH(CH2)SCOOH 3. CH3CH2CH2CH2CH2COOH 4. C6HSCOOCOC6H5 5. CICOCH2CH2CH2CH2OH 6. CH3(CH2)7CONH2 7. CH3(CH2)3CH2COOCH2(CH2).CH3 CH3CH2OCOCH2CH2CH2CH2CH2CH2 8. CH3 9. CH3CH2CH2CH2CH2CH2CH2CH2COCI 10. H2NCOCH2CH2CH2CH3 2.arrow_forward
- 40. Name the following esters b. H3C a. H3C- CH2-CH2 CH2- H3C H3C d. H3C H3C- -CH2 CH2-CH2 CH2- CH2- H2C H2C CH2 CH3 H3C H3C CH3 f. CH-HC HÇ CH2-CH-CH2-CH3 H3C CH2- `CH H2C CH2 H3C h. HCOOCH,CH,CH,CH; CH,CH,COOCH;arrow_forwardProvide reagentsarrow_forward7 8 11 12 me CH3 H3C 3-butylpropanoate 2-butoxypropane propyl-2-butanoate 2-butylpropanoate H₂ C CH3arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning
Nomenclature: Crash Course Chemistry #44; Author: CrashCourse;https://www.youtube.com/watch?v=U7wavimfNFE;License: Standard YouTube License, CC-BY