
(a) Write structural formulas and provide IUPAC names for all the isomeric
(b) Which of the isomers in part (a) yield chiral alcohols on reaction with sodium borohydride?
(c) Which of the isomers in part (a) yield chiral alcohols on reaction with methylmagnesium iodide?
Interpretation:
The given parts are to be explained.
Concept Introduction:
The systematic naming of organic compound is given by IUPAC. The naming of organic compound is done in such a way that the structure of the organic compound is correctly interpreted from its name.
Rules for writing the structural formula from IUPAC are as follows:
First, identify the word root for the given compound.
The suffix used in the compound like
The position and location of the substituents that are bonded to the carbon chain must
identified and number theses substituents on the basis of their priorities.
Answer to Problem 23P
Solution:
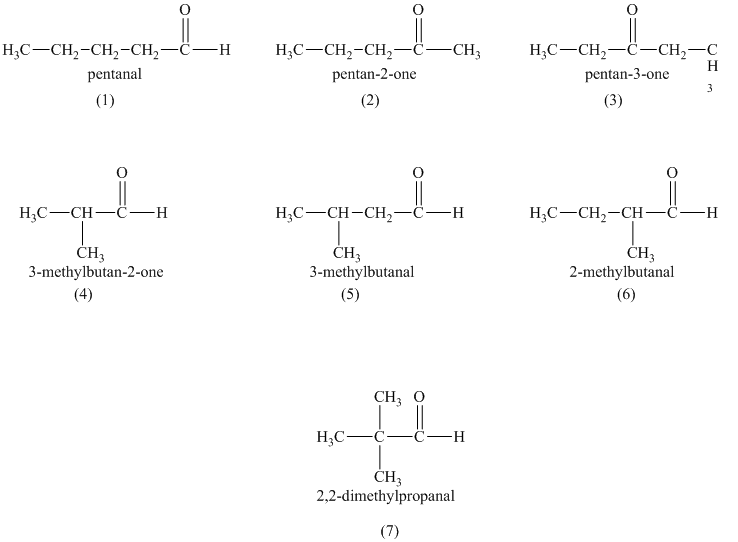
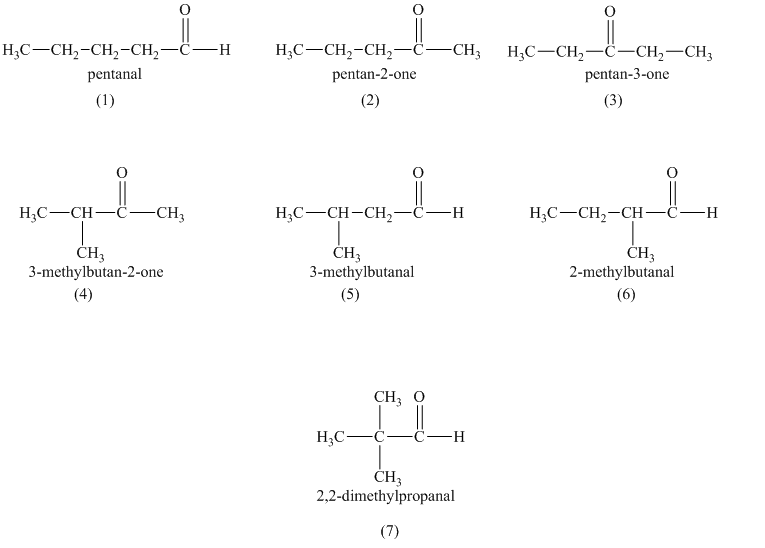
a)
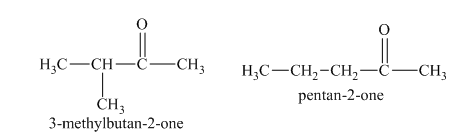
b)
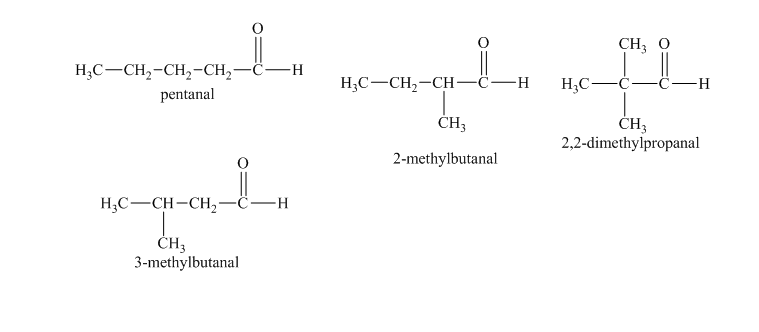
c)
Explanation of Solution
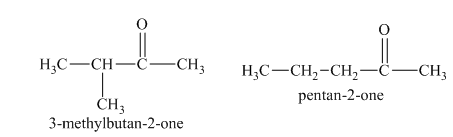
a) Structural formulas and IUPAC name of the isomeric aldehydes and ketones of
The constitutional isomers are the isomers that consist of the same chemical formula but the way they attach to their atoms are totally different from each other.
Aldehydes and ketones contain carbonyl
b) The isomers that form chiral alcohols on the reaction with sodium borohydride.
The reduction of aldehydes with sodium borohydride results in the formation of a primary alcohol, whereas in ketones the formation of secondary alcohols is taken place. Primary alcohols are not chiral in nature. On the other hand, the secondary alcohols that are formed from unsymmetrical ketones show chirality. Therefore, among the isomers in part (a),
c) The isomers that give chiral alcohols on reaction with methylmagnesium iodide.
Methylmagnesium iodide is also known as the Grignard reagent. Aldehydes can give chiral alcohols on reaction with methylmagnesium iodide. The formation of chiral and achiral molecules from the isomers that are present in part(a) are shown as:
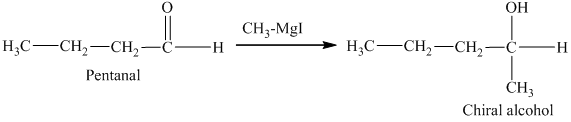
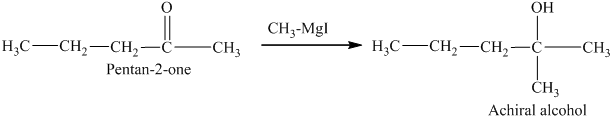
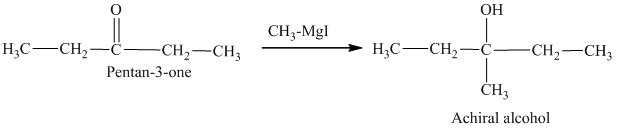
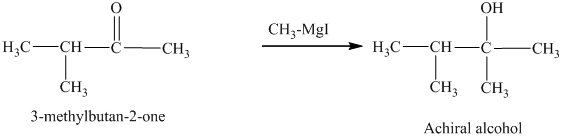
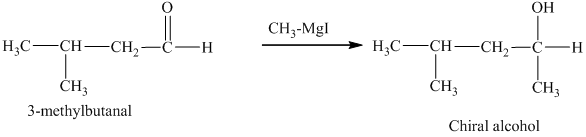
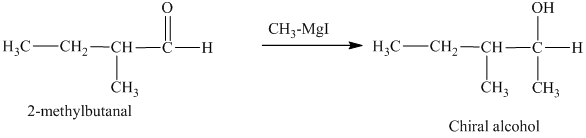
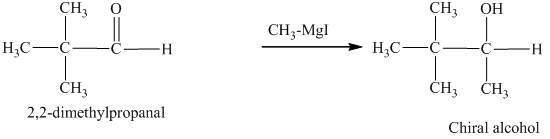
Hence, the isomers that form chiral alcohols on reaction with methylmagnesium iodide are shown below.

Want to see more full solutions like this?
Chapter 18 Solutions
Organic Chemistry - Standalone book
- Compounds B and C are hydrocarbons with the structural formulae as shown below. CH, „CH, в (a) Name compounds B and C according to the IUPAC nomenclature. (b) Both B and C can undergo oxidation reaction with the same oxidizing agent. Write chemical equations involved and explain the differences between these two reactions. (c) Name one reaction that converts B to methylcyclohexane.arrow_forwardGive an IUPAC and common name for each of the following naturally occurring carboxylic acids: (a) CH3CH(OH)CO2H (lactic acid); (b) HOCH2CH2C(OH)(CH3)CH2CO2H (mevalonic acid).arrow_forwardDraw the structure of C4H1,O if the compound: (1) reacts with Na but fails to react with a strong oxidizing agent such as K,Cr,O;; (2) gives a negative iodoform test; and (3) gives a positive Lucas test in 4 minutes. (ii) Give structures and IUPAC names of the alcohols formed from (CH3),CHCH=CH2 by: (i) dilute H,SO,; (ii) В,Нь, then H,Oz, он.arrow_forward
- What is the major organic product of the following reaction? (a) (b) NaBH4 CH3CH₂OH ? (c) (d) OHarrow_forwardWrite structural formulas for all ketones with the molecular formula C6H12O and give each its IUPAC name. Which of these ketones are chiral?arrow_forward(a) A hydrocarbon isolated from fish oil and from plankton was identified as 2,6,10,14-tetramethyl-2-pentadecene. Write its structure.(b) Alkyl isothiocyanates are compounds of the type RN C S. Write a structural formula for allyl isothiocyanate, a pungent-smelling compound isolated from mustard.(c) Grandisol is one component of the sex attractant of the boll weevil. Write a structural formula for grandisol given that R in the structure shown is an isopropenyl group.arrow_forward
- Acetyl chloride, CH3COCl, reacts with the hydroxyl groupsof alcohols to form ester groups with the elimination ofHCl. When an unknown compound X with formulaC4H8O3 reacted with acetyl chloride, a new compound Ywith formula C8H12O5 was formed.(a) How many hydroxyl groups were there in X?(b) Assume that X is an aldehyde. Write a possible structure for X and a possible structure for Y consistent with your structure for X.arrow_forwardDescribe how would you distinguish the following pairs, (a) Benzene and cyclohexane (b) Phenol and toluene (c) Phenol and benzoic acid (d) methanol and isopropyl alcoholarrow_forward4 But-2-enal, CH₂CH=CHCHO, is a pale yellow, flammable liquid with an irritating odour. (a) But-2-enal exists as two stereoisomers. Draw skeletal formulae to show the structure of the two stereoisomers of but-2-enal. (b) (i) Describe a simple chemical test that would show that but-2-enal is an aldehyde. (ii) Explain why this test gives a different result with aldehydes than it does with keton (c) But-2-enal also reacts with sodium borohydride, NaBH4. (i) Identify the organic compound formed in this reaction. (ii) State the type of chemical reaction occurring. (d) Precautions must be taken to prevent but-2-enal catching fire. Construct a balanced equation for the complete combustion of but-2-enal, C₂HO.arrow_forward
- A compound with formula C7H12O is treated with sodium borohydride in methanol to yield 2,2-dimethylcylopentanol. Write a reaction scheme showing the structures of the reactant, the reagents, and the product. Will the product be optically active? Explain.arrow_forwardPredict which member of each group is most soluble in water, and explain the reasons for your predictions.(a) butan-1-ol, pentan-1-ol, or propan-2-ol(b) chlorocyclohexane, cyclohexanol, or cyclohexane-1,2-diol(c) phenol, cyclohexanol, or 4-methylcyclohexanolarrow_forward18. The addition of H20 to ethene yields a molecule called: (a) ethanol (b) ethane (c) ethyne (d) ethylene glycolarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





